Search Results
Showing results 41 to 60 of 96
Invisible Ink
Source Institutions
In this hands-on activity (on page 2 of the PDF), learners experiment with lemon juice and paper to create a message that can only be revealed using chemistry.
Cloudy Globs: Can You Make a White Gel From Two Clear Liquids?
Source Institutions
Using household materials, learners can make white gooey globs from clear solutions. Alum, dissolved in water, reacts with the hydroxide in ammonia to create aluminum hydroxide.
Foam Peanuts
Source Institutions
Learners compare the properties and solubilities of Styrofoam (TM), ecofoam packing peanuts, and popcorn. First, the solubility of each substance is tested in water.

It's a Gas!
Source Institutions
In this activity, learners explore two properties of gases: gases take up space and exert pressure. Learners assemble two flasks and a beaker, connecting them with stoppers and tubing.
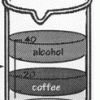
Layers of Liquids
Source Institutions
Learners pour equal amounts of coffee, mineral oil, corn syrup, and alcohol into a beaker. The liquids resolve into stacked layers, and learners can infer which liquids are the most and least dense.

Burning Issues
Source Institutions
Learners use a candle to investigate the products of combustion. When a glass rod is held over a lit candle, the candle flame deposits carbon on the rod.

Curious Contraptions
Source Institutions
In this engineering design activity, learners will design, test, and build a “haunting machine” to solve a Sherlockian mystery.

Balloon in a Flask
Source Institutions
Learners observe a flask with a balloon attached over the mouth and inverted inside the flask.

Crocodiles
Source Institutions
Learners observe and compare the sizes of three toy “growing” crocodiles made from water-absorbent polymers. One is it its original state, dry, hard, and about 10cm long.

Layered Liquids: Chemistry You Can Drink
Source Institutions
In this chemistry activity (on page 2 of the PDF), learners make a layered drink with liquids of different densities.
DNA Extraction: Look at your genes!
Source Institutions
Extract your DNA from your very own cells! First, learners swish salt water in their mouth to collect cheek cells and spit the water into a glass.
A Matter of Splatter
Source Institutions
In this math-based activity, learners will experiment to find how height and angle affect spatter and then use this knowledge to solve a crime.
Mystery Writing: Write and develop a secret message
Source Institutions
Learners write an invisible message using lemon juice on a piece of paper. They then develop the message by soaking the paper in a dilute iodine solution.

What's Your Blood Type?
Source Institutions
In this activity, learners perform a simulated blood test procedure.
Color Me Blue
Source Institutions
In this activity, learners add dilute bleach solution to water that has been dyed with yellow, blue, and green food color.
Of Cabbages and Kings
Source Institutions
This lesson gives full instructions for making cabbage juice indicator, a procedure sheet for learners to record observations as they use the indicator to test materials, and extension activities to d

Trading Places
Source Institutions
In this activity, learners discover that atoms and ions of different metals will change places.

Iron in Cereal: Find iron in your food!
Source Institutions
Learners investigate an iron-fortified cereal by stirring it with a strong magnet. They discover that metallic iron is present in some cereals.

Spicy Indicator: Use turmeric to test for bases in your home
Source Institutions
This activity uses turmeric, a common spice in curry, as an indicator for acidity and basicity. Turmeric is yellow in acid and neutral substances, but turns bright red with bases.

Matter of Degree
Source Institutions
In two separate bags, learners mix water with Epsom salts and detergent.
