Search Results
Showing results 241 to 260 of 284

The Ups and Downs of Thermometers
Source Institutions
In this activity, learners examine the parts of a thermometer. After placing a thermometer in hot and cold water, learners look at molecular model animations of the liquid in a thermometer.

Super Soaker
Source Institutions
In this activity (page 1 of the PDF under SciGirls Activity: Bogs), learners will test cups full of potting soil, sand, and sphagnum moss to see which earth material is able to soak up the most water.

"Boyle-ing" Water
Source Institutions
In this activity, learners explore Boyle's Law and discover that water will boil at room temperature if its pressure is lowered.

Cleaning Water with Dirt
Source Institutions
In this activity on page 7 of the PDF (Water in Our World), learners make their own water treatment systems for cleaning water.

Production of Hydrogen
Source Institutions
In this chemistry activity, learners use mossy zinc (or a galvanized nail) and hydrochloric acid to generate hydrogen gas and test some of its properties.

Recycling Rules: Understanding Recycling and a MRF
Source Institutions
In this activity, learners simulate the separation techniques that materials recovery facilities (MRFs) use and then design their own series of recycling techniques.

Changing the Density of a Liquid: Heating and Cooling
Source Institutions
Learners investigate how the temperature of water affects its density.

Flubber
Source Institutions
Learners experiment with a piece of Silly Putty® by stretching, bouncing, and snapping it. They then create flubber, a similar substance, by mixing diluted glue and a solution of sodium borate.
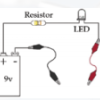
Exploring Materials: Graphene
Source Institutions
In this activity on page 4 of the PDF, explore the unique molecular structure and conductive nature of graphene. Learners construct a circuit with a battery and LED bulb.

Exploring Structures: DNA
Source Institutions
In this activity, learners create a necklace of wheat germ DNA. Learners add alcohol to wheat germ so that the DNA clumps together.

Electroplating
Source Institutions
In this activity, learners electrically plate zinc onto brass objects.

Bury Me Not!
Source Institutions
This activity (page 2 of the PDF under SciGirls Activity: Bogs) is a full inquiry investigation into decomposition.

Stick to It: Adhesion II
Source Institutions
Water sticks to all kinds of things in nature — flowers, leaves, spider webs - and doesn't stick to others, such as a duck's back.

Changing the Density of an Object: Adding Material
Source Institutions
Learners see that a can of regular cola sinks while a can of diet cola floats. As a demonstration, bubble wrap is taped to the can of regular cola to make it float.
Butterflies in Space
Source Institutions
The Butterflies in Space Teacher's Guide uses "life in space" to encourage learners to conduct their own open-ended scientific investigations.

What's in a Penny?
Source Institutions
In this chemistry activity, learners use chemical reactions to observe the composition of an alloy.
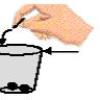
Water Ways
Source Institutions
In this activity (on page 2 of the PDF), learners explore surface tension by adding pennies to cups which are "full" of plain water or soapy water.

Sand Dunes
Source Institutions
This outdoor activity (on page 2 of the PDF under SciGirls Activity: Sand Dunes) is a full inquiry investigation into how the amount of moisture in a sand dune relates to the number of plants growing

I Can't Take the Pressure!
Learners develop an understanding of air pressure in two different activities.

Delicious Smelling Chemistry
Source Institutions
In this activity, learners use household materials to investigate and explore their ability to smell an odor.
