Search Results
Showing results 441 to 460 of 906

Below the Surface: Surface Tension II
Source Institutions
In this activity learners explore surface tension. Why are certain objects able to float on the surface of water and how do detergents break the surface tension of water?
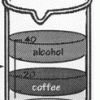
Layers of Liquids
Source Institutions
Learners pour equal amounts of coffee, mineral oil, corn syrup, and alcohol into a beaker. The liquids resolve into stacked layers, and learners can infer which liquids are the most and least dense.

Examining the Heart
Source Institutions
In this activity about the heart (on page 22 of the PDF), learners examine sheep or chicken hearts to learn about the heart's structure and the flow of blood through the heart.

Salt Water Revival
Source Institutions
In this outdoor activity, learners visit the intertidal zone of a rocky coastal site well populated with marine organisms.

Temperature Tactics
Source Institutions
In this activity, learners explore the devices used over time to measure changes in temperature.

Can You Copperplate?
Source Institutions
In this activity, learners explore chemical engineering and how the processes of chemical plating and electroplating have impacted many industries.

The Nose Knows
Source Institutions
In this activity (2nd activity on the page), learners explore how the nose is responsible for part of the flavor we taste in food.

Convection Demonstration
Source Institutions
In this quick activity (located on page 2 of the PDF under GPS: Balloon Fiesta Activity), learners will see the effects of convection and understand what makes hot air balloons rise.

Feel the Heat
Source Institutions
In this design challenge activity, learners design and build a solar hot water heater. Their goal is to create a heater that yields the highest temperature change.

That's the Way the Ball Bounces: Level 2
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, volume, density, and bounce height.

Modeling Tidal Action
Source Institutions
In this activity (Lesson 1), learners work in groups to create tide simulations.

Three Colors of Light
Source Institutions
Have fun with additive mixing! Observe what happens when the three primary colors of light--red, green and blue--are mixed together, resulting in white light.

Composites
Source Institutions
In this activity, learners explore how composites work by creating and testing their own composite for an imaginary company.
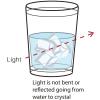
Disappearing Crystals
Source Institutions
Learners experiment with water gel crystals, or sodium polyacrylate crystals, which absorb hundreds of times their weight in water. When in pure water, the water gel crystals cannot be seen.

Design a Submarine
Source Institutions
Learners act as engineers and design mini submarines that move in the water like real submarines.

The China Hammer Mystery
Source Institutions
In this activity, learners are asked to examine the differences between two materials in a pair.

Can Energy be Created or Destroyed?
Source Institutions
In this activity, learners explore conservation of energy by experimenting with a solar cell light device.

Burning Issues
Source Institutions
Learners use a candle to investigate the products of combustion. When a glass rod is held over a lit candle, the candle flame deposits carbon on the rod.

Chocolate (Sea Floor) Lava
Source Institutions
In this edible experiment, learners pour "Magic Shell" chocolate into a glass of cold water. They'll observe as pillow shaped structures form, which resemble lavas on the sea floor.

Observing Different Microbes
Source Institutions
In this activity, learners use a microscope to examine three different microbes: bacteria, yeast and paramecia. Educator will need to prepare the yeast solution one day before the activity.
