Search Results
Showing results 121 to 140 of 154

Cabbage Juice Indicator
Source Institutions
In this chemistry activity, learners make indicator solution from red cabbage. Then, learners test everyday foods and household substances using the cabbage juice indicator.

Automotive Emissions and the Greenhouse Effect
Source Institutions
In this activity about global climate change, learners will conduct an experiment and collect data to compare the amount of carbon dioxide (CO2) in four different sources of gases.

Build a Giant Puzzle!
Source Institutions
In this activity, learners assemble large cubes to make nano-related images. Learners discover how different objects are related to nanoscience and nanotechnology.

Balloon Nanotubes Tabletop
Source Institutions
This activity introduces learners to the structure and properties of carbon nanotubes.
Build A Hydrometer
Source Institutions
In this activity, learners will explore how a hydrometer works by building a working model and conducting experiments.

Turbidity
Source Institutions
This is an activity about turbidity, or the amount of sediment suspended in water.
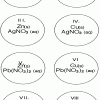
Single Replacement Micro-Reactions
Source Institutions
In this chemistry activity, learners use common chemicals and metals to explore single replacement reactions.

Developing Tests to Distinguish Between Similar-Looking Unknowns
Source Institutions
Learners identify an unknown liquid by comparing its behavior to known liquids. Learners drop liquids onto different surfaces and see how the liquids behave.

Bubbles: Using Controls
In this experiment, learners use JOY liquid detergent and glycerin to make the largest bubble they can that lasts 15 seconds.

Sand, Plants and Pants
Source Institutions
In this activity, learners explore how the application of nano-sized particles or coatings can change a bigger material’s properties.

Soda Geyser
Source Institutions
In this quick activity (page 1 of PDF under SciGirls Activity: Lift Off), learners will use the ever-popular soda geyser experiment to test the reactivity of the various sugar candies or mints.

Monitoring Amphibians
Source Institutions
In this field study, learners discover how to collect data in the field and how their efforts can help certain animals, specifically, amphibians.

Exploring Size: Scented Solutions
Source Institutions
This is an activity in which learners will find that they can detect differences in concentration better with their nose (smelling) than with their eyes (seeing).
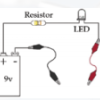
Exploring Materials: Graphene
Source Institutions
In this activity on page 4 of the PDF, explore the unique molecular structure and conductive nature of graphene. Learners construct a circuit with a battery and LED bulb.

Comparing the Amount of Acid in Different Solutions
Source Institutions
In this activity, learners use detergent solution to compare two solutions containing vinegar and cream of tartar.

Temperature Affects Dissolving
Source Institutions
Learners design their own experiment to compare how well cocoa mix dissolves in cold and hot water. They will see that cocoa mix dissolves much better in hot water. Adult supervision recommended.

Bury Me Not!
Source Institutions
This activity (page 2 of the PDF under SciGirls Activity: Bogs) is a full inquiry investigation into decomposition.

Exploring Forces: Gravity
Source Institutions
In this nanoscience activity, learners discover that it's easy to pour water out of a regular-sized cup, but not out of a miniature cup.

Seeds: Roots and Shoots
Source Institutions
In this indepth hands-on activity, learners build a structure that allows them to observe the growth of roots and the correlation between root growth and stem extension.

What's in a Penny?
Source Institutions
In this chemistry activity, learners use chemical reactions to observe the composition of an alloy.
