Search Results
Showing results 321 to 340 of 641

Sweat Spot
Source Institutions
In this activity, learners use a chemical reaction to visualize where moisture forms on the body.

Chemical Methods of Control
Source Institutions
In this lab, learners evaluate the relative effectiveness of various chemical substances (i.e. garlic powder, bathroom cleaner, mouthwash, etc.) as antimicrobial agents.

Build A Battery
Source Institutions
The Let's Do Chemistry "Build a Battery" activity lets participants learn how batteries work and how materials behave, change, and interact by building their own simple battery out of metal and felt w

How Many Pennies?
Source Institutions
In this activity (pages 13-14), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Layered Liquids: Chemistry You Can Drink
Source Institutions
In this chemistry activity (on page 2 of the PDF), learners make a layered drink with liquids of different densities.
DNA Extraction: Look at your genes!
Source Institutions
Extract your DNA from your very own cells! First, learners swish salt water in their mouth to collect cheek cells and spit the water into a glass.

Investigating the Line
Source Institutions
In the related activity called "Colors Collide or Combine," learners are intrigued by the apparent "line" that forms where colors from M&M coatings meet but do not mix.

Biochemistry Happens Inside of You!
Source Institutions
In this four-part activity, learners explore how the body works and the chemistry that happens inside living things.

Soapy Boat
Source Institutions
Learners discover that soap can be used to power a boat. Learners make a simple, flat boat model, put it in water, and then add a drop of detergent at the back of the boat.

We all Scream for Ice Cream
Source Institutions
In this activity, learners observe how salinity affects the freezing point of water by making and enjoying ice cream.
What Does Life Need to Live?
Source Institutions
In this astrobiology activity (on page 11 of the PDF), learners consider what organisms need in order to live (water, nutrients, and energy).
Mystery Writing: Write and develop a secret message
Source Institutions
Learners write an invisible message using lemon juice on a piece of paper. They then develop the message by soaking the paper in a dilute iodine solution.

Polishing Pennies
Source Institutions
In this experiment, learners try different liquids to see which ones clean pennies best. Liquids to try include water, lemon juice, cola, vinegar, and dishwashing detergent.

Self-Portrait Silhouettes: Activity 1
Source Institutions
In this activity, learners make a photographic image--without a camera!
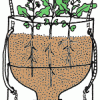
TerrAqua Investigation Column: What is the Land-Water Connection?
Source Institutions
In this investigation, learners plant seeds in a 2-liter bottle filled with soil that is connected to a water source below. Over the next few weeks, learners observe how the plants grow.

Moo Glue
Source Institutions
Using a milk-based recipe, learners create "moo glue" which is basically white school-type glue. The "secret ingredient" in milk that helps make glue is a chemical called casein.
How Do Antacids Work?
Source Institutions
You just ate a big meal and feel heartburn coming on. You take an antacid and feel better. Why? Heartburn is caused by stomach juice (an acid) burning the esophagus.

Fruit Juice Mystery
Source Institutions
In this chemistry challenge, learners work to figure out which of four juices are real, and which is just food coloring and sugar.

Illuminating Luminescence
Source Institutions
In this activity, learners compare and contrast different forms of luminescence by observing how chemiluminescence, phosphorescence, and fluorescence produce or emit light.
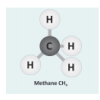
Let's Make Molecules
Source Institutions
In this activity, learners use gumdrops and toothpicks to model the composition and molecular structure of three greenhouse gases: carbon dioxide (CO2), water vapor (H2O) and methane (CH4).
