Search Results
Showing results 181 to 200 of 501

Bernoulli Levitator
Source Institutions
Demonstrate the Bernoulli Principle using simple materials on a small or large scale.

Shrinkers
Source Institutions
In this hands-on activity, learners use heat to shrink samples of polystyrene plastic (#6 recycle code). Learners compare the size and shape of the plastic pieces before and after shrinking.

New Sense about Cents
Source Institutions
In this activity on page 6 of the PDF (Chemistry—It’s Elemental), learners explore some of the properties of copper using a few common household ingredients.

Earthquake Science: Soil Liquefaction
Source Institutions
This activity demonstrates liquefaction, the process by which some soils lose their solidity during an earthquake.

That's the Way the Ball Bounces: Level 1
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, size, and bounce height.

Why is the Sky Blue?
Source Institutions
In this activity, learners use a flashlight, a glass of water, and some milk to examine why the sky is blue and sunsets are red.
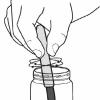
Salting Out
Source Institutions
In this activity, learners create a mixture of water, alcohol and permanent marker ink, and then add salt to form a colored alcohol layer on top of a colorless water layer.
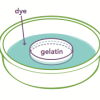
Traveling Nanoparticles Model
Source Institutions
This is an activity (located on page 3 of the PDF under Nanosilver Activity) about diffusion of small molecules across cell membranes.

Moving Molecules!
Source Institutions
In this activity about molecular diffusion (located on page 2 of the PDF under Nanosilver Activity), learners will make predictions and move molecules of iodine through a seemingly solid plastic sandw

Pot-in-Pot Refrigeration
Source Institutions
In this activity (on page 2 of PDF), learners create a low-tech refrigerator that requires no electricity to keep food from spoiling.

What Causes Pressure?
Source Institutions
In this kinesthetic activity that demonstrates pressure, learners act as air molecules in a "container" as defined by a rope.

Diffusion of Water with Gummy Bears
Source Institutions
In this activity, learners investigate the movement of water into and out of a polymer. Learners test the diffusion of water through gummy bears, which are made of sugar and gelatin (a polymer).

From Gas to Liquid to Solid
Source Institutions
What causes frost to form on the outside of a cold container? In this activity, learners discover that liquid water can change states and freeze to become ice.

Lava Lamp
Source Institutions
In this activity, learners will create their own lava lamps. Learners will explore density, effervescence and chemistry through this activity.

Diving Submarine
Source Institutions
Learners use a commercially available toy to experiment with density. They fill a chamber in the toy submarine with baking powder and release it into a tank of water.

A Mole of Gas
Source Institutions
In this two-part activity, learners use everyday materials to visualize one mole of gas or 22.4 liters of gas. The first activity involves sublimating dry ice in large garbage bag.

DIY Bath Bombs
Source Institutions
In this activity, learners will explore acid-base reactions and create their own bubbly results.

Gumdrop Chains and Shrinky Necklaces
Source Institutions
In this activity, learners thread gumdrops together to make a model of a polymer.

Making Sense of Sensors
Source Institutions
In this activity, learners explore sensors and focus specifically on how to measure humidity using a sensor.

Test Density with a Supersaturated Solution
Source Institutions
Learners create three solutions with different levels of salinity. They compare the density of these solutions by coloring them and layering them in a clear plastic cup and in a soda bottle.
