Search Results
Showing results 41 to 58 of 58

How Do Things Fall?
Learners engage in close observation of falling objects. They determine it is the amount of air resistance, not the weight of an object, which determines how quickly an object falls.

What's Your Blood Type?
Source Institutions
In this activity, learners perform a simulated blood test procedure.
Daffy Density
Source Institutions
In this chemistry activity, learners explore density by using four solids and 6 liquids to create colorful, layered rows.

Finding Red
Source Institutions
In this chemistry challenge, learners systematically investigate which combination of four solutions produces a deep red color.

Trading Places
Source Institutions
In this activity, learners discover that atoms and ions of different metals will change places.
Why Are Two Eyes Better Than One?
Source Institutions
In this activity, learners explore how their depth perception would be affected if they only had one eye. Learners work in pairs and attempt to drop a penny in a cup with one eye covered.
Reaction: Yes or No?
Source Institutions
In this activity, learners mix ingredients in a plastic bag, and then identify three characteristics of a chemical reaction: production of heat, color change, and production of a gas.
Drop IT!: Depth Perception
Source Institutions
These two activities (4th on the page) demonstrate the importance of two eyes in judging depth.

Red, White and Blue II Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the rule "likes dissolve likes" by combining three, immiscible liquids to create a colorful density column.
Investigating Convection
Source Institutions
This experiment is designed to illustrate how fluids, including water, have the ability to flow.

Magic Inks
Source Institutions
Learners write their initials by applying different clear "magic ink" solutions to separate pieces of paper and then "develop" the inks with other clear solutions.
Build A Hydrometer
Source Institutions
In this activity, learners will explore how a hydrometer works by building a working model and conducting experiments.

Starch Breakdown
Source Institutions
Learners use Benedict’s solution and heat to test for the presence of simple sugars in glucose, sucrose, starch, and starch combined with amylase.

Witches' Potion Demonstration
Source Institutions
In this chemistry demonstration, learners will discover that phenolphthalein is an acid/base indicator. One learner will read a poem about four witches making a potion.

Exploring Structures: DNA
Source Institutions
In this activity, learners create a necklace of wheat germ DNA. Learners add alcohol to wheat germ so that the DNA clumps together.

Electroplating
Source Institutions
In this activity, learners electrically plate zinc onto brass objects.
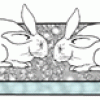
Acid Rain Eats Stone!
Source Institutions
This display shows the dangers of acid rain on buildings and other structures as two concrete bunny rabbits are disintegrated by sulfuric acid. Learners scrape chalk onto the concrete bunnies.

