Search Results
Showing results 41 to 60 of 84
Glitter Slime
Source Institutions
In this activity on page 8 of the PDF, learners make a slimy substance very similar to mucus, and sprinkle it with glitter to imitate the way that allergens are trapped.

Make a Comet Model and Eat It!
Source Institutions
In this activity, learners build models of comets, using edible materials, to learn about comets' structure.

Exploring Properties: Surface Area
Source Institutions
This hands-on activity demonstrates how a material can act differently when it's nanometer-sized.

Red, White and Blue II Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the rule "likes dissolve likes" by combining three, immiscible liquids to create a colorful density column.

Erupting Fizz
Source Institutions
This is a highly visual demonstration that illustrates both the effects of density and chemical reactions.
Squishy Soil
Source Institutions
In this activity, learners will investigate the part of soil. Explore how clay, silt, and sand make up the dirt in your background. Activity includes step-by-step instructions and extension ideas.

Magical Match
Source Institutions
In this demonstration, learners will be "wowed" as three matches burn to form a triangular pyramid shape and "magically" rise off the table.

Gluep
Source Institutions
In this chemistry activity, learners make a slimy non-Newtonian fluid called "Gluep." Use this activity to introduce learners to polymers and viscosity.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.

Building Molecules
Source Institutions
This online interactive has three activities in the NanoLab (press the upper right button): Build, Zoom, and Transform.
Growing Rock Candy
Source Institutions
In this activity, learners make their own rock candy. Crystals will grow from a piece of string hanging in a cup of sugar water. The edible crystals may take up to a week to form.

Gravity and Falling
Source Institutions
This fun and simple hands-on astronomy activity lets learners experiment with a bucket, stretchy fabric, marbles, and weights to discover some basics about gravity.
Shrinkers
Source Institutions
Visitors use heat to shrink samples of polystyrene. They compare samples from containers that were shaped in different ways during manufacturing.

Secret Messages
Source Institutions
In this activity, learners use Teflon (PTFE) tape to write secret messages.
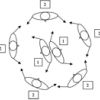
Exploring Fabrication: Self-Assembly
Source Institutions
In this activity, learners participate in several full-body interactive games to model the process of self-assembly in nature and nanotechnology.

Extra Bounce
Source Institutions
In this indoor or outdoor demonstration, use a large and small ball to illustrate conservation of energy and momentum.

Build a Giant Puzzle!
Source Institutions
In this activity, learners assemble large cubes to make nano-related images. Learners discover how different objects are related to nanoscience and nanotechnology.

Balloon Nanotubes Tabletop
Source Institutions
This activity introduces learners to the structure and properties of carbon nanotubes.
