Search Results
Showing results 21 to 40 of 143

Universal Indicator Rainbow Trout
Source Institutions
In this activity on page 2 of the PDF, learners discover how color changes can help scientists distinguish between acids and bases.

Homemade Butter
Source Institutions
In this activity, learners will turn cream and salt into butter—using marbles. Learners will explore how shaking up fat globules help them create homemade butter.

Wild Sourdough
Source Institutions
In this activity, learners explore chemistry and the microbial world by making their own sourdough starter and bread at home using only flour and water.

Instant Ice Cream
Source Institutions
In this activity, learners make instant ice cream without using a freezer.

Solar Powered Cooking
Source Institutions
In this activity, learners make a solar oven. Learners witness the awesome power of the sun to make a yummy treat--a chocolate chip cookie!

Egg Drop
Source Institutions
Perform this classic inertia demonstration to illustrate the transfer of potential energy to kinetic energy.

Oily Ice
Source Institutions
In this activity, learners experiment with the density of ice, water, and oil. Learners will discover that the density of a liquid determines whether it will float above or sink below another liquid.

Egg Osmosis: A four day eggsperience!
Source Institutions
Eggs are placed in vinegar for one or two days to dissolve the shells. Then, learners place the eggs in water or corn syrup and observe them over a period of days.
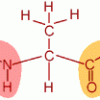
Smell the Maillard Reaction
Source Institutions
In this activity, learners cook amino acids and sugar to explore the range of aromas released.

Light Combinations
Source Institutions
In this activity about magnetism (page 17 of the pdf), learners experiment with magnets, exploring the concept of diamagnetic materials by seeing how a grape reacts to a magnetic field.

Hollandaise Sauce: Emulsion at Work
Source Institutions
In this activity, learners follow a recipe to make hollandaise sauce. Learners discover how cooks use egg yolks to blend oil and water together into a smooth mix.

Yeast Balloons: Can biochemistry blow up a balloon?
Source Institutions
Using yeast, sugar, and water, learners create a chemical reaction which produces carbon dioxide (CO2) gas inside a 2-liter bottle. They use this gas to inflate a balloon.

Can You Make Ice Cream in Two Minutes?
Source Institutions
In this demonstration, learners observe how liquid nitrogen both boils and freezes ingredients to make ice cream in two minutes.

Scream for Ice Cream
Source Institutions
Don't scream for ice cream -- make it with milk, sugar, flavoring and some 'salt-water' ice. Discover the chemistry of ice cream by creating your own.

Root Beer Float
Source Institutions
In this quick activity/demonstration about density, learners examine what happens when two cans of root beer--one diet and one regular--are placed in a large container of water.

ZOOM Glue
Source Institutions
In this activity, learners mix milk, vinegar, baking soda, and water to create sticky glue. Use this activity to explain how engineers develop and evaluate new materials and products.

Supercooled Water Drops
Source Institutions
In this activity, learners touch supercooled water drops with an ice crystal and trigger the water drops to freeze instantly.

Mysterious M&M's
Source Institutions
Learners place an M&M candy in water and observe what happens. The sugar-and-color coating dissolves and spreads out in a circular pattern around the M&M.

Color Changes with Acids and Bases
Source Institutions
Learners mix a variety of substances with red cabbage juice. The juice changes color to indicate whether each substance is an acid or a base.

Making a Battery from a Potato
Source Institutions
In this electrochemistry activity, young learners and adult helpers create a battery from a potato to run a clock.
