Search Results
Showing results 181 to 200 of 277

Catch the (Sound) Wave!
Source Institutions
See and hear how sound waves travel through different types of materials. Extend the experiment online and learn how you can "see" with sound waves using ultrasound.

What Does Spit Do?
Source Institutions
Some animals can swallow food whole, but humans have to chew. In this activity, learners will investigate what saliva does chemically to food before we even swallow.

Drops on a Penny
Source Institutions
In this activity, challenge learners to predict and investigate how many water drops they can fit on one penny.

Make a Comet Model and Eat It!
Source Institutions
In this activity, learners build models of comets, using edible materials, to learn about comets' structure.

Sugar/Salt Crystals
Source Institutions
In this chemistry activity (page 1 of the PDF), learners will observe a physical change.

Static Cling
Source Institutions
In this activity, learners investigate static electricity using everyday objects at four different stations.

Melts in Your Bag, Not in Your Hand
Source Institutions
In this activity, learners use chocolate to explore how the Sun transfers heat to the Earth through radiation.

Gieant Sieve Sorter
Source Institutions
This Exploratorium activity explores size and scale. Through four levels of screen sizes, learners can sort out objects of different sizes.

Red, White and Blue II Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the rule "likes dissolve likes" by combining three, immiscible liquids to create a colorful density column.
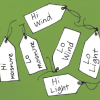
Sensory Hi-Lo Hunt
Source Institutions
In this outdoor activity, learners use only their senses to to find the extremes of several environmental variables or physical factors: wind, temperature, light, slope and moisture.

Finding Colors
Source Institutions
In this chemistry challenge, learners combine acids and bases in a universal indicator to create five different colors.

Ready, Set, Fizz!
Source Institutions
In this activity, learners explore the chemical reaction between water and effervescent antacid tablets. This hands-on activity models how a material can act differently when it's nanometer-sized.
Crystal Garden
Source Institutions
Visitors observe a tray holding a crystal-covered brick. The crystals were created by evaporation of a solution containing liquid bluing, ammonia, and salt.
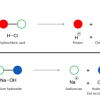
Chemical Identification
Source Institutions
In this activity, learners discover how a cabbage juice indicator helps identify acids and bases, and how iodine indicates the presence of starch.

Disappearing Colors
Source Institutions
In this challenge, learners figure out how to make a juice stain disappear.

Gluep
Source Institutions
In this chemistry activity, learners make a slimy non-Newtonian fluid called "Gluep." Use this activity to introduce learners to polymers and viscosity.

Sunny Day Painting
Source Institutions
In this activity, learners explore properties of water and watch evaporation happen by "painting" with water in the sun.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.

Release the Grease!
Source Institutions
In this simple activity (on page 7 of the PDF), learners use water and liquid dish detergent to see which one removes lipstick better from an index card.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
