Search Results
Showing results 81 to 100 of 123

Copper Cleanup
Source Institutions
In this hands-on experiment, kids use chemistry to explore whether acids or bases are better at restoring a penny’s shine.
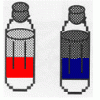
Oil and Soap
Source Institutions
Learners investigate the properties of the liquids in two bottles. One contains layers of oil and water, and one contains oil, water, and soap.

Crystal Stencil Stars
Source Institutions
In this activity on page 6 of the PDF, learners dissolve Epsom salt in water and discover that the resulting solution can be used to create a work of art.

Cauldron Bubbles
Source Institutions
In this activity, learners mix up a bubbly brew and examine density. Learners explore how they can make different materials fall and rise in water using oil, water, and salt.

Using Color to See How Liquids Combine
Source Institutions
Learners add different liquids (water, salt water, alcohol, and detergent solution) to water and observe the different ways the different liquids combine with water.
Let's Go Ice Fishing
Source Institutions
In this activity, learners are challenged to lift a floating ice cube out of a glass of water using just one end of a piece of string.

Matter of Degree
Source Institutions
In two separate bags, learners mix water with Epsom salts and detergent.

Ocean in a Bottle
Source Institutions
In this simulation activity, learners observe what can happen when ocean waves churn up water and oil from an oil spill.

Sugar/Salt Crystals
Source Institutions
In this chemistry activity (page 1 of the PDF), learners will observe a physical change.

Red, White and Blue II Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the rule "likes dissolve likes" by combining three, immiscible liquids to create a colorful density column.

Erupting Fizz
Source Institutions
This is a highly visual demonstration that illustrates both the effects of density and chemical reactions.

Finding Colors
Source Institutions
In this chemistry challenge, learners combine acids and bases in a universal indicator to create five different colors.

Ready, Set, Fizz!
Source Institutions
In this activity, learners explore the chemical reaction between water and effervescent antacid tablets. This hands-on activity models how a material can act differently when it's nanometer-sized.
Big Bubbles
Source Institutions
How do you measure a bubble when it's floating? You can't really, but in this activity, learners can measure the diameter of the ring of suds a bubble leaves on a flat surface.

Crystal Creations: Grow Spikes of Crystals in the Sun
Source Institutions
This activity shows you how to make amazing crystal spikes using Epsom salt and the sun.
Crystal Garden
Source Institutions
Visitors observe a tray holding a crystal-covered brick. The crystals were created by evaporation of a solution containing liquid bluing, ammonia, and salt.
Chemical Identification
Source Institutions
In this activity, learners discover how a cabbage juice indicator helps identify acids and bases, and how iodine indicates the presence of starch.

Disappearing Colors
Source Institutions
In this challenge, learners figure out how to make a juice stain disappear.
Concentrate: Concentrations and Reaction Rates
Source Institutions
Visitors incrementally increase the amount of iodate in three different test tubes containing the same amount of a starch solution.

Carrying Charges: Testing for Conductivity
Source Institutions
Learners are challenged to create solutions that conduct electricity and make a buzzer buzz (or an LED light up).
