Search Results
Showing results 241 to 260 of 327
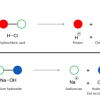
Chemical Identification
Source Institutions
In this activity, learners discover how a cabbage juice indicator helps identify acids and bases, and how iodine indicates the presence of starch.

Disappearing Colors
Source Institutions
In this challenge, learners figure out how to make a juice stain disappear.

Gluep
Source Institutions
In this chemistry activity, learners make a slimy non-Newtonian fluid called "Gluep." Use this activity to introduce learners to polymers and viscosity.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.

Release the Grease!
Source Institutions
In this simple activity (on page 7 of the PDF), learners use water and liquid dish detergent to see which one removes lipstick better from an index card.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.

Using Chemical Change to Identify an Unknown
Source Institutions
In this activity, learners will develop a method to test five similar-looking powders (baking soda, baking powder, cream of tartar, detergent, and cornstarch) with four test liquids (water, vinegar, i

Building Molecules
Source Institutions
This online interactive has three activities in the NanoLab (press the upper right button): Build, Zoom, and Transform.
Growing Rock Candy
Source Institutions
In this activity, learners make their own rock candy. Crystals will grow from a piece of string hanging in a cup of sugar water. The edible crystals may take up to a week to form.

Powder Particulars
Source Institutions
In this introductory activity and demonstration, learners are introduced to the concept that different substances react chemically in characteristic ways.
Shrinkers
Source Institutions
Visitors use heat to shrink samples of polystyrene. They compare samples from containers that were shaped in different ways during manufacturing.
Concentrate: Concentrations and Reaction Rates
Source Institutions
Visitors incrementally increase the amount of iodate in three different test tubes containing the same amount of a starch solution.

Glitter Slime
Source Institutions
Our bodies defend themselves in many different ways to prevent us from getting sick.

Find the Best Way to Clean Oil off Bird Feathers
Source Institutions
In this experiment, learners examine the way oil affects bird feathers and test different cleanup methods to find out which works best.

Secret Messages
Source Institutions
In this activity, learners use Teflon (PTFE) tape to write secret messages.

Carrying Charges: Testing for Conductivity
Source Institutions
Learners are challenged to create solutions that conduct electricity and make a buzzer buzz (or an LED light up).
Exploring Fabrication: Self-Assembly
Source Institutions
In this activity, learners participate in several full-body interactive games to model the process of self-assembly in nature and nanotechnology.

Paint a Fresco
Source Institutions
In this activity on page 7 of the PDF, learners experiment with the interesting designs that can be made by painting on plaster.

Mummy Magic
Source Institutions
Make your own mummy! Using a combination of salts, transform an apple into a mummy and discover how the Ancient Egyptians used drying as one step in the mummification process.
