Search Results
Showing results 181 to 200 of 327

We all Scream for Ice Cream
Source Institutions
In this activity, learners observe how salinity affects the freezing point of water by making and enjoying ice cream.
Mystery Writing: Write and develop a secret message
Source Institutions
Learners write an invisible message using lemon juice on a piece of paper. They then develop the message by soaking the paper in a dilute iodine solution.

Polishing Pennies
Source Institutions
In this experiment, learners try different liquids to see which ones clean pennies best. Liquids to try include water, lemon juice, cola, vinegar, and dishwashing detergent.
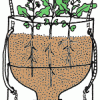
TerrAqua Investigation Column: What is the Land-Water Connection?
Source Institutions
In this investigation, learners plant seeds in a 2-liter bottle filled with soil that is connected to a water source below. Over the next few weeks, learners observe how the plants grow.

Moo Glue
Source Institutions
Using a milk-based recipe, learners create "moo glue" which is basically white school-type glue. The "secret ingredient" in milk that helps make glue is a chemical called casein.
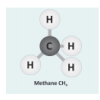
Let's Make Molecules
Source Institutions
In this activity, learners use gumdrops and toothpicks to model the composition and molecular structure of three greenhouse gases: carbon dioxide (CO2), water vapor (H2O) and methane (CH4).

Ice Cream
Source Institutions
In this chemistry activity, learners use the lowered freezing point of water to chill another mixture (ice cream) to the solid state.

Candy Chromatography
Source Institutions
Learners analyze candy-coated sweets using chromatography. Learners use this method to separate the various dyes used to make colored candy.

DIY Elephant Toothpaste
Source Institutions
In this activity, learners will experiment with catalysts to create an at-home version of elephant toothpaste.

Buoyant Bubbles
Source Institutions
What keeps bubbles and other things, like airplanes, floating or flying in the air?

Indicator Paper
Source Institutions
Use grape juice, baking soda, water and vinegar to make acid and base indicator paper! This activity contains a recipe and instructions for the indicator paper.

Chemistry is Colorful
Source Institutions
In this activity, learners explore materials through paper chromatography.

That Sinking Feeling
Source Institutions
In this quick activity, learners observe how salinity and temperature affect the density of water, to better understand the Great Ocean Conveyor.
Daffy Density
Source Institutions
In this chemistry activity, learners explore density by using four solids and 6 liquids to create colorful, layered rows.

Rain Machine (Solar Still)
Source Institutions
In this activity, learners work in groups to build simple solar stills filled with salt water. After the stills are complete, learners observe what happens when they place the stills in the sun.

Mystery Sand
Source Institutions
In this activity, learners play with surprising sand that doesn’t get wet! Learners explore how water behaves differently when it comes in contact with "magic sand" and regular sand.

Kool Colors
Source Institutions
Learn about dyes and mordants (fixatives) when you tie-dye fabric with Kool-Aid™ and vinegar. The colored molecules in Kool-Aid™ form a chemical bond between the fiber and dye molecules.
Of Cabbages and Kings
Source Institutions
This lesson gives full instructions for making cabbage juice indicator, a procedure sheet for learners to record observations as they use the indicator to test materials, and extension activities to d
Making Recycled Paper
Source Institutions
In this activity on page 11 of the PDF, learners follow simple steps to recycle old newspaper into new paper.

Racing M&M Colors
Source Institutions
Learners design their own experiment to determine which M&M color dissolves the fastest in water.
