Search Results
Showing results 81 to 100 of 126

Conversation Piece
Source Institutions
Focus sound through a balloon! In this Exploratorium activity, you'll use dry ice to create a balloon that's a sound lens.

Condiment Diver
Source Institutions
In this hands-on activity, learners make the world's simplest Cartesian diver, using only a plastic bottle, some water, and a condiment packet.
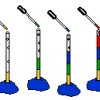
Liquid Layers
Source Institutions
Experiment with liquids of different densities and create liquid layers. For example, oil and water have different densities: oil floats on water because it is less dense than water.
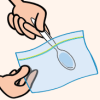
Glitter Slime
Source Institutions
In this activity on page 8 of the PDF, learners make a slimy substance very similar to mucus, and sprinkle it with glitter to imitate the way that allergens are trapped.

Using Color to See How Liquids Combine
Source Institutions
Learners add different liquids (water, salt water, alcohol, and detergent solution) to water and observe the different ways the different liquids combine with water.
Let's Go Ice Fishing
Source Institutions
In this activity, learners are challenged to lift a floating ice cube out of a glass of water using just one end of a piece of string.

Super Shrinkers
Source Institutions
In this activity on page 14 of the PDF (Rethinking the 3 R’s: It’s Easy to be Green), learners turn plain pieces of recycled plastic into shrunken works of art.

Drops on a Penny
Source Institutions
In this activity, challenge learners to predict and investigate how many water drops they can fit on one penny.

Light as Air
Source Institutions
In this physics activity (page 6 of the PDF), learners will demonstrate air has weight by comparing an inflated balloon to a deflated one.

Make a Comet Model and Eat It!
Source Institutions
In this activity, learners build models of comets, using edible materials, to learn about comets' structure.

Exploring Properties: Surface Area
Source Institutions
This hands-on activity demonstrates how a material can act differently when it's nanometer-sized.

Salt Crystal Garden
Source Institutions
In this activity, learners will explore saturated solutions and discover how crystals form.

Red, White and Blue II Demonstration
Source Institutions
In this chemistry demonstration, learners investigate the rule "likes dissolve likes" by combining three, immiscible liquids to create a colorful density column.

Erupting Fizz
Source Institutions
This is a highly visual demonstration that illustrates both the effects of density and chemical reactions.

Tissue Paper Spray Art
Source Institutions
In this activity, learners observe color mixing and absorbency using colored tissue paper and spray bottles.

Gluep
Source Institutions
In this chemistry activity, learners make a slimy non-Newtonian fluid called "Gluep." Use this activity to introduce learners to polymers and viscosity.

Sunny Day Painting
Source Institutions
In this activity, learners explore properties of water and watch evaporation happen by "painting" with water in the sun.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.
