Search Results
Showing results 1 to 19 of 19

Liquid Body Armor
Source Institutions
In this activity, learners explore how nanotechnology is being used to create new types of protective fabrics.

Fused Plastic Wallet
Source Institutions
In this up-cycling activity, learners recycle plastic bags and repurpose them into useful wallets.

Gummy Shapes
Source Institutions
In this activity, learners use chemistry to “self-assemble” gummy shapes. Learners discover that self-assembly is a process by which molecules and cells form themselves into functional structures.

Great Balls of Goop
Source Institutions
In this activity, learners mix white craft glue and borax solution together to produce a surprising new material: GOOP!
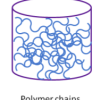
Polymers are Chains (K-2)
Source Institutions
In this activity, learners make a paper model of a polymer, then make Silly Putty, an actual polymer.
Snow Day!
Source Institutions
In this activity (on pages 4-5), learners make fake snow by adding water to the super-absorbant chemical from diapers, sodium polyacrylate.

The Unpoppable Balloon
Source Institutions
In this activity, learners explore polymer structure and their ability to reform around objects by attempting to stab a wooden skewer through a balloon without popping it.

What Counts in Bounce
Source Institutions
In this activity learners compare the bounciness of warm and cold racquetballs to see if temperature makes a difference in how well they bounce.

Gumdrop Chains and Shrinky Necklaces
Source Institutions
In this activity, learners thread gumdrops together to make a model of a polymer.

Shrinkers: Cook up some plastic!
Source Institutions
In this activity (on page 2 of the PDF), learners (with adult help and supervision) investigate how heat affects polystyrene plastic.
Choose Your Ooze
Source Institutions
During this activity, learners will make different versions of "ooze" using varied proportions of detergent and glue.
Glitter Slime
Source Institutions
In this activity on page 8 of the PDF, learners make a slimy substance very similar to mucus, and sprinkle it with glitter to imitate the way that allergens are trapped.

Spaghetti Strength
Source Institutions
In this activity on page 7 of the PDF, learners explore how engineers characterize building materials.
Fantastic Plastic
Source Institutions
In this activity, learners investigate the properties of plastic bags. Learners find out what happens when they slowly push a pencil through a plastic bag filled with water.

Exploring A Hydrogel
Source Institutions
In this activity on page 10 of the PDF, learners develop an experiment to answer the following question: "How much water can the hydrogel in a baby diaper hold?" Use this activity to explore polymers,

Exploring Fabrication: Gummy Capsules
Source Institutions
In this activity, learners make self-assembled polymer spheres.

Squidgy Slime
Source Institutions
In this chemistry activity, learners transform two ingredients (4% polyvinyl alcohol solution and 4% borax solution) into gooey slime.

Slime
Source Institutions
Learners make slime from white school glue and Borax detergent. The long chain molecules of the white glue become cross-linked by the Borax into a big network.

Flubber: Make a polymer!
Source Institutions
This activity (on page 2 of the PDF) features a recipe to create the stretchy polymer Flubber from Borax detergent, white glue, and water.
