Search Results
Showing results 581 to 600 of 796
Crystal Garden
Source Institutions
Visitors observe a tray holding a crystal-covered brick. The crystals were created by evaporation of a solution containing liquid bluing, ammonia, and salt.

Breathing Blue
Source Institutions
In this activity, learners test exhaled breath for carbon dioxide and learn how to use an indicator as a simple way to measure pH.

Disappearing Colors
Source Institutions
In this challenge, learners figure out how to make a juice stain disappear.

Moisture Makers
Source Institutions
In this outdoor activity, learners compare the moisture released from different kinds of leaves and from different parts of the same leaf, by observing the color change of cobalt chloride paper.

Wind Turbine Blade Design
Source Institutions
In this activity, learners design, build and test wind turbines. Learners go through the design process and use the scientific method to test important blade variables.

Gluep
Source Institutions
In this chemistry activity, learners make a slimy non-Newtonian fluid called "Gluep." Use this activity to introduce learners to polymers and viscosity.

Jumping Beans
Source Institutions
In this physics activity (page 3 of the PDF), learners will explore the concept of static electricity.
Light and Sound
Source Institutions
In this four-part activity, learners explore light and sound through a variety of hands-on investigations.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.

Release the Grease!
Source Institutions
In this simple activity (on page 7 of the PDF), learners use water and liquid dish detergent to see which one removes lipstick better from an index card.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.

Tug-of-War
Source Institutions
This activity (on page 2 of the PDF under SciGirls Activity: Tug O' War) is a full inquiry investigation into tug-of-war physics. Groups of learners will test two tug-of-war strategies.

Using Chemical Change to Identify an Unknown
Source Institutions
In this activity, learners will develop a method to test five similar-looking powders (baking soda, baking powder, cream of tartar, detergent, and cornstarch) with four test liquids (water, vinegar, i
Are you a Supertaster?
Source Institutions
In this activity, learners examine their tongue and taste buds.

Density Intensity
Source Institutions
In this activity on page 12 of the PDF (Rethinking the 3 R’s: It’s Easy to be Green), learners examine how recyclable materials are separated by various properties at recycling centers.

Foam Rocket
Source Institutions
In this activity, learners work in teams build and launch rubberband-powered foam rockets.
Growing Rock Candy
Source Institutions
In this activity, learners make their own rock candy. Crystals will grow from a piece of string hanging in a cup of sugar water. The edible crystals may take up to a week to form.
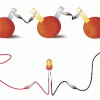
Electricity: Fruit Batteries
Source Institutions
In this activity, learners create a battery from fruit. This activity helps learners explore electricity, electrochemistry, and series circuits as well as the process of scientific inquiry.

Introduction to the Scientific Method
Source Institutions
In this activity (page 26 of the PDF), learners make observations, formulate hypotheses and design a controlled experiment, based on the reaction of carbon dioxide with calcium hydroxide.
