Search Results
Showing results 421 to 440 of 573

Gluep
Source Institutions
In this chemistry activity, learners make a slimy non-Newtonian fluid called "Gluep." Use this activity to introduce learners to polymers and viscosity.
Light and Sound
Source Institutions
In this four-part activity, learners explore light and sound through a variety of hands-on investigations.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.

Release the Grease!
Source Institutions
In this simple activity (on page 7 of the PDF), learners use water and liquid dish detergent to see which one removes lipstick better from an index card.
Air-filled (Pneumatic) Bone Experiments
Source Institutions
Just like birds, some dinosaurs had air-filled (pneumatic) bones, which made the dinosaurs' skeletons lighter.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.

Using Chemical Change to Identify an Unknown
Source Institutions
In this activity, learners will develop a method to test five similar-looking powders (baking soda, baking powder, cream of tartar, detergent, and cornstarch) with four test liquids (water, vinegar, i

Density Intensity
Source Institutions
In this activity on page 12 of the PDF (Rethinking the 3 R’s: It’s Easy to be Green), learners examine how recyclable materials are separated by various properties at recycling centers.

Building Molecules
Source Institutions
This online interactive has three activities in the NanoLab (press the upper right button): Build, Zoom, and Transform.
Growing Rock Candy
Source Institutions
In this activity, learners make their own rock candy. Crystals will grow from a piece of string hanging in a cup of sugar water. The edible crystals may take up to a week to form.
The Nose Knows!
Source Institutions
In this activity on page 9 of the PDF, learners test how flavoring extracts move through the walls of a balloon.
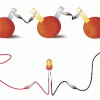
Electricity: Fruit Batteries
Source Institutions
In this activity, learners create a battery from fruit. This activity helps learners explore electricity, electrochemistry, and series circuits as well as the process of scientific inquiry.

Introduction to the Scientific Method
Source Institutions
In this activity (page 26 of the PDF), learners make observations, formulate hypotheses and design a controlled experiment, based on the reaction of carbon dioxide with calcium hydroxide.
Density Rainbow
Source Institutions
In this activity, learners mix several sugar solutions to investigate the property of density. Each sugar solution has a different density and color of the rainbow.

pHun with Cabbage
Source Institutions
In this chemistry activity, learners will test the pH of various foods and household substances using cabbage.

Protein Bracelets
Source Institutions
In this activity, learners use beads, which represent amino acids, to create protein bracelets. Learners examine the relationship between amino acids and proteins.

Paper making: a craft and a chemical engineering major
Source Institutions
In this activity, learners explore the question "What is paper?" Learners discover the processes and materials required to make paper while experimenting with different recycled fibers and tools.

Taking Its Temperature
Source Institutions
In this activity (pages 5-7), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Geyser
Source Institutions
This Exploratorium activity can be used in many contexts because geysers are great opportunities for learning about heat and temperature changes as well as geological/space science phenomena.
