Search Results
Showing results 101 to 120 of 126
Finding the Carbon in Sugar
Source Institutions
In this activity about combustion and energy, learners observe a burning candle in a sealed jar and the burning of white sugar.

Save Your Skin
Source Institutions
This is a fun activity about the power of the Sun and the importance of using sunscreen to protect your sensitive skin from its rays.

Frog Eggs
Source Institutions
In this activity, learners compare frog eggs to chicken eggs to better understand why frog eggs need water. Learners compare a boiled chicken egg to "frog eggs" represented by boiled tapioca.
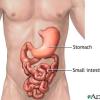
Ziploc Digestion Simulator
Source Institutions
In this biology activity, learners recreate the process of digestion in a zip lock bag. A bit of soda pop added to some crumbled crackers approximates how acids in the stomach dissolve food.

Iodine Investigators!
Source Institutions
In this activity on page 7 of the PDF (Chemistry—It’s Elemental), learners use iodine to identify foods that contain starch.

Kimchee Fermentation Chamber
Source Institutions
Learners make kimchee or sauerkraut, which is really just fermented cabbage, in a 2-liter plastic bottle.

Starch Breakdown
Source Institutions
Learners use Benedict’s solution and heat to test for the presence of simple sugars in glucose, sucrose, starch, and starch combined with amylase.

Testing Vitamin C: Chemistry's Clear Solution
Source Institutions
In this activity on page 8 of the PDF, learners investigate vitamin C. Learners conduct a chemistry experiment to determine if Tang drink mix or orange juice contains more vitamin C.

Homework, Hogwarts Style
Source Institutions
In this activity on page 8 of the PDF (Behind the Scenes with Chemistry), learners make three of Harry Potter's essential school supplies: quills, ink, and color-changing paper.

Make Your Own Soda Pop
Source Institutions
In this chemistry activity (page 8 of the PDF), learners will identify the instances of physical change, chemical change, and solutions while making homemade soda pop.

Exploring Structures: DNA
Source Institutions
In this activity, learners create a necklace of wheat germ DNA. Learners add alcohol to wheat germ so that the DNA clumps together.

Bury Me Not!
Source Institutions
This activity (page 2 of the PDF under SciGirls Activity: Bogs) is a full inquiry investigation into decomposition.

I Can't Take the Pressure!
Learners develop an understanding of air pressure in two different activities.

Delicious Smelling Chemistry
Source Institutions
In this activity, learners use household materials to investigate and explore their ability to smell an odor.

Try Growing Your Own Mold
Source Institutions
This is a hands-on activity that uses bread and household materials to grow mold. Learners collect dust from a room, wipe it on food, and contain it. One to seven days later, mold has grown.

Fungus Among Us
Source Institutions
In this environmental health activity, learners grow and observe bread mold and other kinds of common fungi over the course of 3-7 days.

Wintergreen
Source Institutions
In this outdoor, winter activity, learners find living green plants under the snow and determine the light and temperature conditions around the plants.

Microbes are Everywhere
Source Institutions
In this four-day activity, learners grow bacteria and/or fungi from a variety of locations and compare the results.

Cabbage Patch Chemistry
Source Institutions
In this chemistry activity, learners will learn how to make their own pH indicator using cabbage leaves, and then test common household items with their homemade indicator.

