Search Results
Showing results 141 to 160 of 387

New Sense about Cents
Source Institutions
In this activity on page 6 of the PDF (Chemistry—It’s Elemental), learners explore some of the properties of copper using a few common household ingredients.

Wet Art
Source Institutions
In this activity (located on page 10 of the PDF), learners explore the properties of spraying and dripping water, while making art.
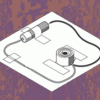
Hidden Alarm
Source Institutions
In this design challenge activity, learners invent a device that will make their friends and family ask, "What's buzzing?" Learners design an alarm with an on/off switch that is small enough to hide.

What does Color have to do with Cooling?
Source Institutions
In this demonstration/experiment, learners discover that different colors and materials (metals, fabrics, paints) radiate different amounts of energy and therefore, cool at different rates.

Magnetic Pendulum
Source Institutions
In this activity about magnetism (page 15 of the PDF), learners will explore how opposite and similar magnetic poles affect a swinging (pendulum) magnet.

Penny Cup Game
Source Institutions
In this optics activity, learners conduct an experiment to find out why two eyes are better than one!

Chemical Breath
Source Institutions
This is a chemistry lab activity about solutions (page 7 of the PDF). Learners see firsthand how chemicals in a solution can combine to form an entirely different substance.

Peripheral Vision
Source Institutions
In this optics activity, learners conduct an experiment to explore peripheral vision. Learners collect data about their ability to see shapes, colors, or letters using their peripheral vision.

Moving Molecules!
Source Institutions
In this activity about molecular diffusion (located on page 2 of the PDF under Nanosilver Activity), learners will make predictions and move molecules of iodine through a seemingly solid plastic sandw

Paper Bridge
Source Institutions
In this engineering activity, challenge learners to construct a paper bridge that can support 100 pennies.
Cooling the Mummy's Tomb
Source Institutions
In this activity, learners conduct an experiment to help Pharaoh design a better insulated tomb.

From Gas to Liquid to Solid
Source Institutions
What causes frost to form on the outside of a cold container? In this activity, learners discover that liquid water can change states and freeze to become ice.

¡Espirales de Colores!
Source Institutions
En esta actividad sobre la densidad de los líquidos, los participantes ven si las gotas de colorante se hunden en diferentes soluciones de agua.

A Mole of Gas
Source Institutions
In this two-part activity, learners use everyday materials to visualize one mole of gas or 22.4 liters of gas. The first activity involves sublimating dry ice in large garbage bag.

Disappearing Glass Rods
Source Institutions
In this optics activity, learners discover how they can make glass objects "disappear." Learners submerge glass objects like stirring rods into a beaker of Wesson™ oil to explore how the principles of

Using Tools and Melting Ice
Source Institutions
In this activity, learners will experiment with different materials that can melt and change ice.
Catapult
Source Institutions
In this activity about transfer of energy, learners get to build their own catapult using nothing more than a plastic spoon and some tape.

Power Up!
Source Institutions
In this online game, learners must purchase power plants for their city.

Hanford at the Half-Life Radiation Calculator
Source Institutions
This quiz lets you estimate your annual radiation exposure.

Mixing and Unmixing in the Kitchen
Source Institutions
In this chemistry investigation, learners combine common cooking substances (flour, baking powder, sugar, salt, pepper, oil, water, food coloring) to explore mixtures.
