Search Results
Showing results 21 to 32 of 32

Liquid Crystal Thermometers
Source Institutions
In this activity, learners explore liquid crystal thermometers to observe how heat flows by conduction, convection, radiation, and evaporation.
Reaction: Yes or No?
Source Institutions
In this activity, learners mix ingredients in a plastic bag, and then identify three characteristics of a chemical reaction: production of heat, color change, and production of a gas.
Sodium Acetate Hand Warmers
Source Institutions
In this activity, sodium acetate hand warmers are used to introduce learners to supersaturated solutions, crystallization, and exothermic reactions.
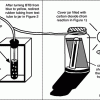
How Greenhouse Gases Absorb Heat
Source Institutions
Learners observe two model atmospheres -- one with normal atmospheric composition and another with an elevated concentration of carbon dioxide.

Shape Up!
Source Institutions
In this activity (pages 8-9), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Taking Its Temperature
Source Institutions
In this activity (pages 5-7), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Hot Stuff!
Source Institutions
In this activity, learners discover that sand is the major ingredient in glass.

Hot & Cold
Source Institutions
In this activity, learners experiment with hydrogen peroxide, vinegar, yeast, and baking soda to produce hot and cold reactions. Use this activity to demonstrate exothermic and endothermic reactions.

Air Pressure
Source Institutions
In this experiment, learners use a blow dryer and water bottle to observe and record changes in air pressure caused by changes in temperature.

Global Climate Change and Sea Level Rise
Source Institutions
In this activity, learners practice the steps involved in a scientific investigation while learning why ice formations on land (not those on water) will cause a rise in sea level upon melting.

Having a Gas with Water
Source Institutions
In this activity, learners construct a simple electrolysis device. With this device, learners can decompose water into its elemental components: hydrogen and oxygen gas.

That's the Way the Ball Bounces: Level 3
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, volume, density, and bounce height.
