Search Results
Showing results 181 to 192 of 192

Global Climate Change and Sea Level Rise
Source Institutions
In this activity, learners practice the steps involved in a scientific investigation while learning why ice formations on land (not those on water) will cause a rise in sea level upon melting.

Milk Plastic
Source Institutions
In this activity, learners transform everyday milk into small plastic figurines and jewelry. Use this activity to introduce learners to monomers and polymers.

Wintergreen
Source Institutions
In this outdoor, winter activity, learners find living green plants under the snow and determine the light and temperature conditions around the plants.
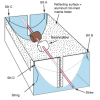
Reflective Solar Cooker
Source Institutions
In this activity, learners use the Sun's energy to cook marshmallows. Learners construct the solar oven out of simple everyday materials.

Having a Gas with Water
Source Institutions
In this activity, learners construct a simple electrolysis device. With this device, learners can decompose water into its elemental components: hydrogen and oxygen gas.
Light Bulb Challenge
Source Institutions
In this activity, learners explore the difference between compact fluorescent light (CFL) bulbs and traditional incandescent bulbs.

Change in Temperature: Exothermic Reaction
Source Institutions
Learners add calcium chloride to a baking soda solution and observe an increase in temperature along with the production of a gas and a white precipitate. These are all signs of a chemical reaction.

Backyard Biodiesel
Source Institutions
In this activity, learners make a small batch of biodiesel that will work in any diesel engine. Learners use an old juice bottle as a "reactor" vessel to chemically process vegetable oil into fuel.

What's in the Water
Source Institutions
"What's in the Water" lets participants use tools to solve the mystery- what chemicals and compounds are in a sample of water?

That's the Way the Ball Bounces: Level 3
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, volume, density, and bounce height.

Hot Spoons
Source Institutions
In this physics activity (page 2 of the PDF), learners will explore the concept of thermal conductivity and learn the differences between high and low thermal conductivity.

Keep it Cool
Source Institutions
In this activity, learners explore how engineers have met the challenge of keeping foods, liquids, and other items cool.
