Search Results
Showing results 81 to 100 of 117

Super Shrinkers
Source Institutions
In this activity on page 14 of the PDF (Rethinking the 3 R’s: It’s Easy to be Green), learners turn plain pieces of recycled plastic into shrunken works of art.

Sugar Crystal Challenge
Source Institutions
This lesson focuses on surface area and how the shape of sugar crystals may differ as they are grown from sugars of different coarseness.
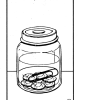
Reaction: Yes or No?
Source Institutions
In this activity, learners mix ingredients in a plastic bag, and then identify three characteristics of a chemical reaction: production of heat, color change, and production of a gas.
Sodium Acetate Hand Warmers
Source Institutions
In this activity, sodium acetate hand warmers are used to introduce learners to supersaturated solutions, crystallization, and exothermic reactions.
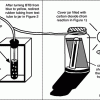
How Greenhouse Gases Absorb Heat
Source Institutions
Learners observe two model atmospheres -- one with normal atmospheric composition and another with an elevated concentration of carbon dioxide.

Make a Comet Model and Eat It!
Source Institutions
In this activity, learners build models of comets, using edible materials, to learn about comets' structure.

Sugar/Salt Crystals
Source Institutions
In this chemistry activity (page 1 of the PDF), learners will observe a physical change.

Shape Up!
Source Institutions
In this activity (pages 8-9), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.
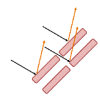
Liquid Crystals Interact with Light!
Source Institutions
In this two-part activity, learners explore the properties of liquid crystals, which are responsible for why mood rings change color.

Wind Power: Creating a Wind Generator
Source Institutions
This lesson challenges groups of learners to design and construct a wind generator with the most electrical output.
Investigating Convection
Source Institutions
This experiment is designed to illustrate how fluids, including water, have the ability to flow.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.

Taking Its Temperature
Source Institutions
In this activity (pages 5-7), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

Geyser
Source Institutions
This Exploratorium activity can be used in many contexts because geysers are great opportunities for learning about heat and temperature changes as well as geological/space science phenomena.

Make a Salt Volcano (Lava Lite)
Source Institutions
This activity about density provides instructions for making a miniature "lava lite" with just salt, oil, water, and food coloring.

The Rain Man
Source Institutions
In this activity, learners observe the hydrologic cycle in action as water evaporates and condenses to form rain right before their eyes.

Density Rainbows
Source Institutions
In this activity, learners explore the concept of density by pouring 5 different liquids into a jar. Food coloring is added if needed to give each liquid a distinct color.

Hot Stuff!
Source Institutions
In this activity, learners discover that sand is the major ingredient in glass.

Milli's Insulation Investigation
Source Institutions
In this activity on page 2 of the PDF, learners test different materials to find out which is the best insulator.

Hot & Cold
Source Institutions
In this activity, learners experiment with hydrogen peroxide, vinegar, yeast, and baking soda to produce hot and cold reactions. Use this activity to demonstrate exothermic and endothermic reactions.
