Search Results
Showing results 61 to 79 of 79
Finding the Carbon in Sugar
Source Institutions
In this activity about combustion and energy, learners observe a burning candle in a sealed jar and the burning of white sugar.

Moving On Up: Capillary Action 1
Source Institutions
Over the course of several days, learners explore the property of water that helps plants move water from roots to leaves or gives paper towels the capacity to soak up water.

Battling for Oxygen
Working in groups, learners model the continuous destruction and creation of ozone (O3) molecules, which occur in the ozone layer.

Automotive Emissions and the Greenhouse Effect
Source Institutions
In this activity about global climate change, learners will conduct an experiment and collect data to compare the amount of carbon dioxide (CO2) in four different sources of gases.

Build a Giant Puzzle!
Source Institutions
In this activity, learners assemble large cubes to make nano-related images. Learners discover how different objects are related to nanoscience and nanotechnology.

Iodine Investigators!
Source Institutions
In this activity on page 7 of the PDF (Chemistry—It’s Elemental), learners use iodine to identify foods that contain starch.

Hand Battery
Source Institutions
In this activity about chemistry and electricity, learners form a battery by placing their hands onto plates of different metals.
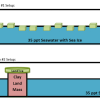
Causes and Effects of Melting Ice
Source Institutions
In this activity, learners explore the concept of density-driven currents (thermohaline circulation) and how these currents are affected by climate change.

Sand, Plants and Pants
Source Institutions
In this activity, learners explore how the application of nano-sized particles or coatings can change a bigger material’s properties.

Exploring Size: Scented Solutions
Source Institutions
This is an activity in which learners will find that they can detect differences in concentration better with their nose (smelling) than with their eyes (seeing).

Exploring Structures: DNA
Source Institutions
In this activity, learners create a necklace of wheat germ DNA. Learners add alcohol to wheat germ so that the DNA clumps together.

Bury Me Not!
Source Institutions
This activity (page 2 of the PDF under SciGirls Activity: Bogs) is a full inquiry investigation into decomposition.

Off Base
Source Institutions
In this activity, learners explore the factors that tend to resist changes in pH of the ocean and why the ocean is becoming more acidic.

Delicious Smelling Chemistry
Source Institutions
In this activity, learners use household materials to investigate and explore their ability to smell an odor.

Biomimicry: Synthetic Gecko Tape Through Nanomolding
Source Institutions
In this activity/demo, learners examine a synthetic gecko tape with micron sized hairs that mimics the behavior of the gecko foot.

I Spy Nano!
Source Institutions
In this game, learners try to find nano-related objects on a game board. Learners investigate the different ways nano is in the world around us.
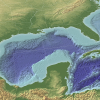
The Dead Zone: A Marine Horror Story
Source Institutions
In this environmental science and data analysis activity, learners work in groups to track a Dead Zone (decreased dissolved oxygen content of a body of water) using water quality data from the Nutrien

Forces at the Nanoscale: Nano Properties of Everyday Plants
Source Institutions
This is an activity (located on page 3 of PDF under Nasturtium Leaves Activity) about surface tension.

Self-Assembly Game
Source Institutions
This is a quick game about self-assembly (page 2 of PDF under Self-Assembly Activity). Like the molecules of DNA, learners will self-assemble into a pattern by following a simple set of rules.
