Search Results
Showing results 681 to 700 of 799

Iodine Investigators!
Source Institutions
In this activity on page 7 of the PDF (Chemistry—It’s Elemental), learners use iodine to identify foods that contain starch.

Exploring A Hydrogel
Source Institutions
In this activity on page 10 of the PDF, learners develop an experiment to answer the following question: "How much water can the hydrogel in a baby diaper hold?" Use this activity to explore polymers,

Hand Battery
Source Institutions
In this activity about chemistry and electricity, learners form a battery by placing their hands onto plates of different metals.

Exploring Fabrication: Gummy Capsules
Source Institutions
In this activity, learners make self-assembled polymer spheres.

Developing Tests to Distinguish Between Similar-Looking Unknowns
Source Institutions
Learners identify an unknown liquid by comparing its behavior to known liquids. Learners drop liquids onto different surfaces and see how the liquids behave.

Seeing Is Believing
Source Institutions
This activity is designed to accompany the PBS documentary about African-American chemist "Percy Julian: Forgotten Genius." Learners look through two cups with small holes in them to simulate what it'

Kimchee Fermentation Chamber
Source Institutions
Learners make kimchee or sauerkraut, which is really just fermented cabbage, in a 2-liter plastic bottle.

Bubbles: Using Controls
In this experiment, learners use JOY liquid detergent and glycerin to make the largest bubble they can that lasts 15 seconds.

Starch Slime
Source Institutions
Learners mix liquid water with solid cornstarch. They investigate the slime produced, which has properties of both a solid and a liquid.

The Ups and Downs of Thermometers
Source Institutions
In this activity, learners examine the parts of a thermometer. After placing a thermometer in hot and cold water, learners look at molecular model animations of the liquid in a thermometer.

Solving Dissolving
Source Institutions
The Sacred Cenote at Chichén Itzá is a sink hole, or well, containing groundwater. In this activity, learners create their own cenote using chalk, limestone, acids, and rain water.
Create a Mangrove Tree
Source Institutions
In this group activity, learners will explore the characteristics, functions and uniqueness of the mangrove tree.

Floating and Falling Flows
Learners create beautiful fluid motion. They explore fluid dynamics, surface tension, solubility, and buoyancy while mixing liquids together.

Starch Breakdown
Source Institutions
Learners use Benedict’s solution and heat to test for the presence of simple sugars in glucose, sucrose, starch, and starch combined with amylase.

Breaking Up with Combustion
Source Institutions
This activity teaches combustion as the interaction of a fuel source and oxygen.

Four of the States of Matter
Source Institutions
This kinesthetic science demonstration introduces learners to four states of matter: solid, liquid, gas, and plasma.

Dissolving Different Liquids in Water
Source Institutions
In this activity, learners add different liquids to water and apply their working definition of “dissolving” to their observations.
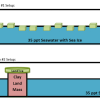
Causes and Effects of Melting Ice
Source Institutions
In this activity, learners explore the concept of density-driven currents (thermohaline circulation) and how these currents are affected by climate change.

"Boyle-ing" Water
Source Institutions
In this activity, learners explore Boyle's Law and discover that water will boil at room temperature if its pressure is lowered.

Conservation of Mass
Source Institutions
This activity was designed for blind learners, but all types of learners can participate to learn about conservation of gas. This is one of the classic experiments using baking soda and vinegar.
