Search Results
Showing results 781 to 799 of 799
Dinosaur Bone Experiments
Source Institutions
This activity features two connected hands-on activities about dinosaur bones.
Balanced Budget Chemistry
Source Institutions
In this activity, learners balance chemical equations and discover the law of conservation of mass. Learners use coins to model molecules to balance the equations.

Backyard Biodiesel
Source Institutions
In this activity, learners make a small batch of biodiesel that will work in any diesel engine. Learners use an old juice bottle as a "reactor" vessel to chemically process vegetable oil into fuel.

Sublimation Bubbles
Source Institutions
"Sublimation Bubbles" allows learners to explore how some solid materials, such as dry ice, can phase change directly from their solid to gaseous form.

What's in the Water
Source Institutions
"What's in the Water" lets participants use tools to solve the mystery- what chemicals and compounds are in a sample of water?

Make a Water Cycle Wristband
Source Institutions
In this activity, learners thread colored beads onto string. Each beach represent a process of the water cycle.

Production of a Gas: Controlling a Chemical Reaction
Source Institutions
Learners mix vinegar and baking soda to produce a gas. With the addition of a bit of liquid soap, the gas becomes trapped in measurable bubbles.

That's the Way the Ball Bounces: Level 3
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, volume, density, and bounce height.

Self-Assembly Game
Source Institutions
This is a quick game about self-assembly (page 2 of PDF under Self-Assembly Activity). Like the molecules of DNA, learners will self-assemble into a pattern by following a simple set of rules.

Flubber: Make a polymer!
Source Institutions
This activity (on page 2 of the PDF) features a recipe to create the stretchy polymer Flubber from Borax detergent, white glue, and water.

The Senses of "Unknown Creatures"
Source Institutions
In this activity, learners use earthworms as "unknown creatures" from the South American jungle to find out how animals use senses.

Outbreak!: Investigating Epidemics
Source Institutions
A group of learners simulates the spread of disease by trading slips of paper, one set of which has been treated with a baking soda solution.

Plaster Casts
Source Institutions
In this activity, learners combine two substances (plaster of Paris and water) to make a cast of an object's imprint in clay.

Cabbage Chemistry
Source Institutions
In this chemistry activity (page 5 of the PDF), learners make an acid-base indicator using cabbage. Learners then explore how various subtances react with this indicator.
Sticky Situation
Source Institutions
In this activity, learners separate the protein from milk and and use it to make their own glue.

Paper Whites
Source Institutions
Learners observe different paper samples under ordinary room light and under a black light to learn some of the chemical differences between different types of paper.

Keep it Cool
Source Institutions
In this activity, learners explore how engineers have met the challenge of keeping foods, liquids, and other items cool.

Waterproof that Roof!
Source Institutions
In this activity, learners explore how engineers have improved roofing designs and materials in order to protect the contents of buildings.
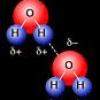
Cohesion Coin
Source Institutions
In this activity about the property of water (page 6 of the PDF), learners use a coin to demonstrate cohesion between water molecules, exploring the molecular forces that allow water molecules to "
