Source Institutions
Source Institutions
Add to list Go to activity
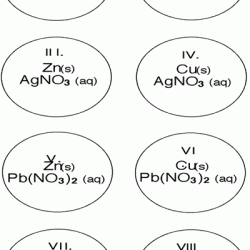
In this chemistry activity, learners use common chemicals and metals to explore single replacement reactions. Learners will record any color changes or gas production for each reaction as well as write a balanced equation for each.
- Under 5 minutes
- 10 to 30 minutes
- $1 - $5 per student
- Ages 11 - 18
- Activity, Experiment/Lab Activity
- English
Quick Guide
Materials List (per student)
- zinc or galvanized nail
- aluminum foil
- copper or copper wire
- 0.1 M CuSO4
- 0.1 M AgNO3
- 0.1 M ZnSO4
- 1 M HCl
- 8 watch glasses or a spot plate
- 8 pipets
Subjects
-
Mathematics
-
Data Analysis and Probability
- Data Analysis
- Data Collection
-
Data Analysis and Probability
-
Physical Sciences
-
Chemistry
- Chemical Reactions
- Acids and Bases
-
States of Matter
- Gases
-
Structure and Properties of Matter
- Elements and Periodic Table
-
Chemistry
-
The Nature of Science
-
The Scientific Process
- Conducting Investigations
- Gathering Data
- Formulating Explanations
- Communicating Results
-
The Scientific Process
Audience
To use this activity, learners need to:
- see
- see color
- read
- touch
Learning styles supported:
- Involves hands-on or lab activities
Other
This resource is part of:
Access Rights:
- Free access with registration
