Search Results
Showing results 1261 to 1280 of 1480

Exploring Structures: Butterfly
Source Institutions
In this activity, learners investigate how some butterfly wings get their color.

Look Mom, No Wings!
Source Institutions
In this activity about flight, learners explore how high they can jump. Learners dip their finger in ink or dirt, then jump as high as they can and mark paper attached to the wall.

How the Mushroom Got Its Spots
Source Institutions
In this activity (p.26 of PDF), learners discover why mushrooms have spots. Learners use a balloon, toilet paper, and water to simulate what happens as mushrooms grow.

Dissolving a Substance in Different Liquids
Source Institutions
In this activity, learners make colored sugar and add it to water, alcohol, and oil to discover some interesting differences in dissolving.

Unlocking the Secret in Product Codes
Source Institutions
In this activity, learners will explore how product barcodes are scanned accurately and be able to "guess" the last number in a 13 digit barcode correctly every time.

Frog Olympics
Source Institutions
Did you know that a bullfrog can jump a distance of 10 times its body length? Learn more about nature's most acrobatic amphibian, the frog, through this set of short, hands-on activities.
Invisible Investigations
Source Institutions
Using indirect observational methods, learners distinguish between charged and uncharged objects.

Special Effects Using Household Chemicals
Source Institutions
In this activity on page 4 of the PDF (Behind the Scenes with Chemistry), learners make some special effects, including snow and breaking glass, with supplies found in the home.

Dancing Cereal
Source Institutions
In this quick activity (on page 2 of the PDF under GPS: Body Electricity Activity), learners will observe how dry breakfast cereal appears to dance when it gets close to a balloon charged with static

Making Sodium Acetate: Hot Ice
Source Institutions
In this chemistry activity which should only be done under adult supervision (page 10 of the PDF), learners will create an exothermic process by making Sodium Acetate.

Super Soaking Materials
Source Institutions
In this activity, learners will test cups full of potting soil, sand, and sphagnum moss to see which earth material is able to soak up the most water.

The World's Water
Source Institutions
Water on Earth is in lakes, the ocean, rivers, underground, and frozen glaciers.
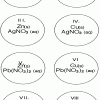
Single Replacement Micro-Reactions
Source Institutions
In this chemistry activity, learners use common chemicals and metals to explore single replacement reactions.

Drip, Drop, Drip, Drop
Source Institutions
In this math lesson, learners design an experiment to model a leaky faucet and determine the amount of water wasted due to the leak.

Hot Stuff!: Investigation #3
Learners test two jars of ice water, one covered and one open, for changes in temperature. After placing the jars in the sun, learners discover that the covered jar cools down more slowly.

Do Sweat It!
Source Institutions
In this activity, learners explore why humans sweat. Learners compare the effects of heat on a balloon filled with air and a balloon filled water.

Challenge: Microgravity
Source Institutions
In this activity about the circulatory system and space travel (on page 38 of the PDF), learners use water balloons to simulate the effects of gravity and microgravity on fluid distribution in the bod

Stained Glass Glue
Source Institutions
In this activity on page 6 of the PDF, learners use glue instead of glass to create artwork that can be hung in a window.

Marble Challenge
Source Institutions
In this physics activity (page 6 of the PDF), learners will try to keep a marble in a bottomless cup without touching the marble inside.

Iodine Investigators!
Source Institutions
In this activity on page 7 of the PDF (Chemistry—It’s Elemental), learners use iodine to identify foods that contain starch.
