Search Results
Showing results 601 to 620 of 641

Try Growing Your Own Mold
Source Institutions
This is a hands-on activity that uses bread and household materials to grow mold. Learners collect dust from a room, wipe it on food, and contain it. One to seven days later, mold has grown.

The Power of Graphene
Source Institutions
This lesson focuses on graphene and its electrical properties and applications.

Fireworks in a Glass
Source Institutions
In this activity, learners use water, oil, and food coloring to observe a chemical reaction that creates a shower of colors inside of a glass.

Stuck on You: Adhesion
Source Institutions
Learners explore water adhesion and learn about why water molecules are more strongly attracted to some substances than others.

All Mixed Up!
Source Institutions
In this activity, learners separate a mixture of pebbles, salt crystals, and wood pieces. They add water and pour the mixture through a strainer.
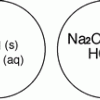
Gas Producing Micro-Reaction
Source Institutions
In this chemistry activity, learners use common chemicals and metals to examine reactions that produce gaseous substances.
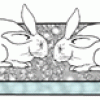
Acid Rain Eats Stone!
Source Institutions
This display shows the dangers of acid rain on buildings and other structures as two concrete bunny rabbits are disintegrated by sulfuric acid. Learners scrape chalk onto the concrete bunnies.

Squidgy Slime
Source Institutions
In this chemistry activity, learners transform two ingredients (4% polyvinyl alcohol solution and 4% borax solution) into gooey slime.

Sticky Engineering Challenge
Source Institutions
In this activity, learners explore how engineers work in a team to solve problems.
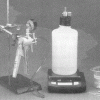
As Light as Air
Source Institutions
Learners measure a bottle full of air, and then use a vacuum pump to remove the air. When they re-weigh the bottle, learners find the mass is about 0.8g less.

Milk Plastic
Source Institutions
In this activity, learners transform everyday milk into small plastic figurines and jewelry. Use this activity to introduce learners to monomers and polymers.

Shake the Bag Ice Cream
Source Institutions
In this activity, learners will experiment with salt and ice in order to turn a bag of ingredients into ice cream.

Levity Through Tension: Fun with Water's Surface Tension
Source Institutions
This experiment describes how to create a "dribble bottle" which only leaks water when the cap is unscrewed. The full water bottle has a small hole made with a push pin.

Chemistry Makes Scents
Source Institutions
In "Chemistry Makes Scents," participants use their noses to distinguish between chemicals with very similar structures.

Pages of a Forbidden Tome
Source Institutions
In this activity, learners use chemistry to produce weathered "antiqued" paper with burned edges. Learners first soak white paper in coffee and then apply a charring solution of ammonium chloride.

See the Colors in Leaves
Source Institutions
Learners use chromatography to separate and analyze the mixture of pigments in leaves. Use this activity to discuss photosynthesis as well as why leaves change color in autumn.

Dye Detective
Source Institutions
Learners use filter paper and water to analyze six different markers. They mark the paper with ink, and dip the paper in water. The water travels up the paper and dissolved ink travels with it.

Forces at the Nanoscale: Nano Properties of Everyday Plants
Source Institutions
This is an activity (located on page 3 of PDF under Nasturtium Leaves Activity) about surface tension.

Change in Temperature: Exothermic Reaction
Source Institutions
Learners add calcium chloride to a baking soda solution and observe an increase in temperature along with the production of a gas and a white precipitate. These are all signs of a chemical reaction.

Burst a Bubble
Source Institutions
In this activity, learners will create their own bubble solution. Learners will explore chemistry, geometry and trial and error through this activity.
