Search Results
Showing results 21 to 40 of 49

Rock Bottoms
Source Institutions
Learners add acid rain (nitric acid) to two cups that represent lakes. One cup contains limestone gravel and the other contains granite gravel.

Food Forensics: A Case of Mistaken Identity
Source Institutions
This lesson is designed to serve as an introduction to the immune system. It can stand alone or it can lead into further studies of the immune system.

Float Your Boat
Source Institutions
In this physics activity, learners will explore buoyancy.

Testing Antimicrobials:: Antibacterial Soap? Do They All Work the Same?
Source Institutions
Many common household cleaners are antimicrobials.
Masses & Springs
Source Institutions
In this online activity, learners use a realistic mass and spring laboratory. They hang masses from springs and adjust the spring stiffness and damping.

Invisible Sunblock
Source Institutions
This is a hands-on activity exploring how nanoscale particles are used in mineral sunblocks to increase their transparency.

Demonstrating An Epidemic
Source Institutions
This experiment allows learners to experience a small scale "epidemic," demonstrating the ease with which disease organisms are spread, and enables learners to determine the originator of the "epidemi
Ionic Bonding Puzzle Lab
Source Institutions
In this activity, learners create models of ionic compounds and observe the chemical formula of binary molecules they have created.
Stretching Wires
Source Institutions
In this activity, learners determine the elastic and plastic properties of different types of metal wires.

Corals on Acid
Source Institutions
The objective of this inquiry-based lesson is for learners to gain an understanding of how increasing ocean acidity can affect the calcification of marine organisms.

Ready, Set, Fizz!
Source Institutions
In this activity, learners explore the chemical reaction between water and effervescent antacid tablets. This hands-on activity models how a material can act differently when it's nanometer-sized.

Home Mycology Lab
Source Institutions
Agriculturalists have long considered mushroom growing a challenge, largely because you need a piece of benchtop equipment known as a laminar flow hood.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.
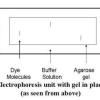
Gel Electrophoresis of Dyes
Source Institutions
In this experiment related to plant biotechnology, learners discover how to prepare and load an electrophoresis gel.

Taking Its Temperature
Source Institutions
In this activity (pages 5-7), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

What's the Matter
Source Institutions
In this activity, learners identify different classes of matter based on physical properties.

Lotus Leaf Effect
Source Institutions
This is a demonstration about how nature inspires nanotechnology. It is easily adapted into a hands-on activity for an individual or groups.
Concentrate: Concentrations and Reaction Rates
Source Institutions
Visitors incrementally increase the amount of iodate in three different test tubes containing the same amount of a starch solution.
