Search Results
Showing results 41 to 60 of 123

Disappearing Statues
Source Institutions
In this activity (on page 8), learners model how marble statues and buildings are affected by acid rain.

Hot and Cold
Source Institutions
In this activity, learners explore temperature changes from chemical reactions by mixing urea with water in one flask and mixing calcium chloride with water in another flask.

New Sense about Cents
Source Institutions
In this activity on page 6 of the PDF (Chemistry—It’s Elemental), learners explore some of the properties of copper using a few common household ingredients.

Limewater
Source Institutions
This is a chemistry lab activity about solutions (page 6 of the PDF). Students make a limewater testing solution for carbon dioxide and explore the concepts of solubility and precipitates.

Full of Hot Air: Hot Air Balloon Building
Source Institutions
In this activity, learners create a model of a hot air balloon using tissue paper and a hairdryer. Educators can use this activity to introduce learners to density and its role in why things float.

See the Light
Source Institutions
Learners mix a solution of luminol with hydrogen peroxide to produce a reaction that gives off blue light.

Tempest in a Teacup
Source Institutions
In this hands-on activity, learners determine the types of chemical reactions achieved when combining different household products.

Mixing and Unmixing in the Kitchen
Source Institutions
In this chemistry investigation, learners combine common cooking substances (flour, baking powder, sugar, salt, pepper, oil, water, food coloring) to explore mixtures.

Comparing Crystals
Source Institutions
In this chemistry activity (page 3 of the PDF), learners will learn about crystals by growing their very own.
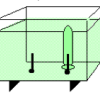
Electrolysis
Source Institutions
Using electrolysis, learners produce hydrogen gas and oxygen gas from water molecules in a solution.

Cabbage Juice Indicator: Test the pH of household products
Source Institutions
Learners make their own acid-base indicator from red cabbage. They use this indicator to test substances around the house.

Miscibility
Source Institutions
Learners observe a bottle containing water and oil. They are invited to pick up the bottle and mix the contents together.

COSI Quicksand
Source Institutions
In this chemistry experiment, learners get to make a very bizarre substance using corn starch and water. Is it a solid? Is it a liquid? Or is it a different kind of substance entirely?
Inner Space
Source Institutions
In this activity, learners discover that there is space between molecules even in a cup "full" of water. They first fill a cup with marbles, and then add sand to fill the gaps between the marbles.

Color Splash
Source Institutions
In this activity, learners mix water, cooking oil, and liquid food coloring to create beautiful colored designs in a cup. Use this activity to explore liquid density and solubility.
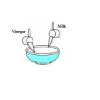
Plastic Milk: You can make plastic from milk
Source Institutions
In this activity (on page 2 of the PDF), learners make a plastic protein polymer from milk. Adding vinegar to milk causes the protein casein to solidify or curdle.

Rock Candy
Source Institutions
In this yummy chemistry activity which requires adult supervision, learners use sugar and water to explore how crystals form.

Nature of Dye
Source Institutions
"Nature of Dye" allows participants to create their own dyes and art while exploring how chemicals interact and how these interactions can have real-world applications.

COSI's Bubble Recipe
Source Institutions
Everybody loves bubbles, and this is the best bubble recipe ever!

Pepper Scatter
Source Institutions
In this quick activity, learners break the tension that happens when water develops a "skin." Learners use water, pepper and some soap to discover the wonders of surface tension—the force that attract
