Search Results
Showing results 241 to 260 of 660

Egg Bungee Jump
Source Institutions
In this activity, learners design a bungee jump for an egg using nylon stockings.

Avogadro's Bubbly Adventure
Source Institutions
In this activity on page 7 of the PDF, learners investigate the solubility of gas in water at different temperatures. This experiment will help learners determine if temperature affects solubility.

Tempest in a Teacup
Source Institutions
In this hands-on activity, learners determine the types of chemical reactions achieved when combining different household products.
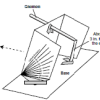
Using a Sundial
Source Institutions
In this activity (on page 12 of the PDF), learners make a sundial (shadow clock) appropriate for their geographic location in the northern hemisphere and use it to tell time.

To Dye For
Source Institutions
Learners add two dyes to mineral oil and water, and then compare their miscibility (how well they mix) in each.

Measuring the Wind
Source Institutions
In this activity, learners explore how anemometers work to record wind speeds and how the equipment has undergone engineering adaptations over time.

Sidewalk Chalk
Source Institutions
In this chemistry activity, learners witness an exothermic reaction, while making their very own, completely usable sidewalk chalk. This is also an excellent activity for exploring color mixing.

Super Sleuths
Source Institutions
In this physical sciences activity, learners use science to solve a "crime." Learners collect trace evidence (glitter) and explore its characteristics, such as color, size, shape, and light reflection

Acids & Bases
Source Institutions
In this activity, learners test the pH of safe liquids available at home by creating a pH indicator from mashed blueberries.

Egg Bungee
Source Institutions
Learners attach an egg to a rubber-band bungee cord and drop the egg.

Edible Glass
Source Institutions
In this activity, learners discover the principles of edible glass by making a supersaturated sugar solution.

Nanoparticle Stained Glass
Source Institutions
In this activity/demo, learners are introduced to the connection between medieval stained glass artisans and nanotechnology.

Science of Sunblock
Source Institutions
This is an activity (located on page 3 of the PDF under Stained Glass Activity) about nanotechnology making its way into everyday products, such as sunscreen, and how effective these invisible particl

Comparing Crystals
Source Institutions
In this chemistry activity (page 3 of the PDF), learners will learn about crystals by growing their very own.

Sink It
Source Institutions
Learners classify a variety of objects by their characteristics. They then design an experiment to determine which objects float or sink in water and add this characteristic to their classification.

Seas in Motion
Source Institutions
In this outdoor, beach activity, learners use tennis balls, water balloons and other simple devices to investigate the movement of waves and currents off a sandy beach.
Chemical Reactions in Your Mouth
Source Institutions
In this chemistry activity (page 5 of the PDF), learners will see that chewing is more than just the crushing up of food; there is actually a chemical change going on at the same time.

Temperature vs. Height: Soda Geyser Series #6
Source Institutions
In this activity, learners conduct a controlled experiment to examine how temperature will affect the height of a soda geyser.

Half Full or Half Empty
Source Institutions
In this activity (12th activity on the page), learners conduct an experiment to demonstrate how muscles are constantly feeding information to the brain about what they are doing.
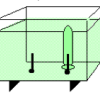
Electrolysis
Source Institutions
Using electrolysis, learners produce hydrogen gas and oxygen gas from water molecules in a solution.
