Search Results
Showing results 241 to 260 of 573

Molecules in Motion
Source Institutions
"Molecules in Motion" explores how materials behave and change in a vacuum.

Unleakable Bag
Source Institutions
In this activity, learners explore the flexibility of polymer chains by pushing sharpened pencils through a Ziploc bag.

Instant Ice Cream with a Dry Ice Bath
Source Institutions
In this chemistry meets cooking activity, learners make carbonated, vanilla ice cream using dry ice and denatured ethanol, which are both inexpensive and accessible.
Take Out the Trash
Source Institutions
Learners explore how recyclers take advantage of the different properties of materials, such as magnetism and density, to separate them from a mixture.

Crystal Painting
Source Institutions
In this activity, learners will "paint" their own crystal artwork by creating a picture with a super saturated salt solution.

The Carbon Cycle and its Role in Climate Change: Activity 2
Source Institutions
In this activity (on page 7), learners explore the meaning of a "carbon sink." Using simple props, learners and/or an educator demonstrate how plants act as carbon sinks and how greenhouse gases cause

Why Does Food Spoil?
Source Institutions
In this activity, learners will conduct an experiment to discover methods of reventing foot mold growth on food.

Lost Labels
Source Institutions
In this experiment, learners will conduct chemical and physical tests to identify mystery substances.

Water "Digs" It!
Source Institutions
In this activity, learners investigate soil erosion. Learners set up a simulation to observe how water can change the land and move nutrients from one place to another.
Inner Space
Source Institutions
In this activity, learners discover that there is space between molecules even in a cup "full" of water. They first fill a cup with marbles, and then add sand to fill the gaps between the marbles.

Color Splash
Source Institutions
In this activity, learners mix water, cooking oil, and liquid food coloring to create beautiful colored designs in a cup. Use this activity to explore liquid density and solubility.

Design a Flavor: Experiment to Make Your Own Ice Cream Flavor!
Source Institutions
In this delicious activity, learners get to make, taste-test and compare their own "brands" of homemade strawberry ice cream.
Plastic Milk: You can make plastic from milk
Source Institutions
In this activity (on page 2 of the PDF), learners make a plastic protein polymer from milk. Adding vinegar to milk causes the protein casein to solidify or curdle.
Invisible Ink
Source Institutions
In this hands-on activity (on page 2 of the PDF), learners experiment with lemon juice and paper to create a message that can only be revealed using chemistry.

Energy For Life
Source Institutions
In this activity about the relationship between food and energy (page 1 of PDF), learners observe and quantify the growth of yeast when it is given table sugar as a food source.
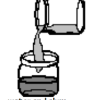
Cloudy Globs: Can You Make a White Gel From Two Clear Liquids?
Source Institutions
Using household materials, learners can make white gooey globs from clear solutions. Alum, dissolved in water, reacts with the hydroxide in ammonia to create aluminum hydroxide.

Rock Candy
Source Institutions
In this yummy chemistry activity which requires adult supervision, learners use sugar and water to explore how crystals form.

Nature of Dye
Source Institutions
"Nature of Dye" allows participants to create their own dyes and art while exploring how chemicals interact and how these interactions can have real-world applications.

Tiny Geyser Models
Source Institutions
In this activity (located on page 2), learners will construct tiny model geysers out of film canisters, warm water, and antacid seltzer tablets.

Pop Rockets
Source Institutions
Learners place water and part of an antacid tablet in a film canister. The reaction creates a gas reaction that launches the film canister like a rocket.
