Search Results
Showing results 1 to 18 of 18

Electroplating
Source Institutions
In this electrochemistry activity, learners will explore two examples of electroplating.

Conductivity Meter
Source Institutions
In this activity, learners build a simple qualitative conductivity tester with a battery, bulb and foil.

Aluminum-Air Battery: Foiled again!
Source Institutions
Construct a simple battery that's able to power a small light or motor out of foil, salt water, and charcoal. A helpful video, produced by the Exploratorium, guides you along on this activity.

Making a Battery from a Potato
Source Institutions
In this electrochemistry activity, young learners and adult helpers create a battery from a potato to run a clock.

Glow Up
Source Institutions
In this activity, learners explore chemiluminescence and fluorescence. Learners examine 3 different solutions in regular light, in the dark with added bleach solution, and under a black light.

Pickle-oh!: Musical Pickle Instrument
Source Institutions
What's a Pickle-Oh? Two pieces of pickle on a stick are connected to a Pico Cricket (micro controller). When you slide the pickles apart the note changes.
Neural Network Signals
Source Institutions
In this activity, learners create an electrical circuit and investigate how some dissolved substances conduct electricity.

Make Your Own Batteries!
Source Institutions
This activity (on page 3 of the PDF under GPS: Body Electricity Activity) is a full inquiry investigation into conductivity.

Harvesting Chemicals from a Battery
Source Institutions
In this activity, learners take apart a used zinc-carbon dry cell battery.

Circuit Bending with Play-Doh
Source Institutions
Break open that used musical toy and squish some Play-Doh over the circuit boards, and you will hear some weird and distorted sounds the manufacturer never intended!

Cake by Conduction
Source Institutions
In this demonstration, cook a cake using the heat produced when the cake batter conducts an electric current.

Indicating Electrolysis
Source Institutions
In this activity, learners build a simple electrolysis device. Then learners use an indicating solution to visualize hydrogen and oxygen molecules in water.
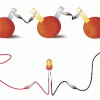
Electricity: Fruit Batteries
Source Institutions
In this activity, learners create a battery from fruit. This activity helps learners explore electricity, electrochemistry, and series circuits as well as the process of scientific inquiry.

Penny Battery
Source Institutions
In this activity, learners light an LED with five cents. Learners use two different metals and some sour, salty water to create a cheap battery.

Carrying Charges: Testing for Conductivity
Source Institutions
Learners are challenged to create solutions that conduct electricity and make a buzzer buzz (or an LED light up).

Photolithography
Source Institutions
In this activity, learners use UV light to transfer a pattern onto a plastic board. The pattern is transferred by placing a mask (a transparency sheet with the pattern) on a plastic board.

The Power of Graphene
Source Institutions
This lesson focuses on graphene and its electrical properties and applications.

Having a Gas with Water
Source Institutions
In this activity, learners construct a simple electrolysis device. With this device, learners can decompose water into its elemental components: hydrogen and oxygen gas.
