Search Results
Showing results 61 to 80 of 146

Acids & Bases
Source Institutions
In this activity, learners test the pH of safe liquids available at home by creating a pH indicator from mashed blueberries.

Mixing and Unmixing in the Kitchen
Source Institutions
In this chemistry investigation, learners combine common cooking substances (flour, baking powder, sugar, salt, pepper, oil, water, food coloring) to explore mixtures.
Piece It Together: Puzzle Hunt
Source Institutions
In this activity, learners follow clues to find five puzzle pieces, then assemble them. This activity works well with a whole group, individuals, or families.

Enzyme Action
Source Institutions
In this activity that can be used as a lab or demonstration, learners use Lactaid® and lactose to demonstrate the concept of enzyme action.

Mighty Molecules
Source Institutions
In this activity, learners use marshmallows and gum drops to construct seven models of molecules. Learners classify (solid, liquid or gas) and draw diagrams of the molecules.

Tasty Buds
Source Institutions
In this activity (1st activity on the page), learners explore their sense of taste and the structure of the tongue by taste-testing various foods.

Cabbage Juice Indicator: Test the pH of household products
Source Institutions
Learners make their own acid-base indicator from red cabbage. They use this indicator to test substances around the house.

In Proportion
Source Institutions
Through this nutrition activity (page 5 of the PDF), learners will understand—and probably be surprised by—how big serving sizes of various foods should be.

Atoms and Matter (K-2)
Source Institutions
In this activity, learners explore atoms as the smallest building blocks of matter. With adult help, learners start by dividing play dough in half, over and over again.
Invisible Ink
Source Institutions
In this hands-on activity (on page 2 of the PDF), learners experiment with lemon juice and paper to create a message that can only be revealed using chemistry.

A Slime By Any Other Name
Source Institutions
This fun video explains how to make a batch of oobleck (or slime) and why this special substance is known as a "non-Newtonian" fluid. Watch as Mr.

What's the Difference between Weather and Climate?
Source Institutions
In this interactive and informative group activity, learners use packages of M&M's to illustrate the difference between weather and climate.

Chocoberry Chillers: Volume for Thought
Source Institutions
In this Cyberchase activity, have fun measuring volumes and enjoy a delicious shake.
Many Seeds: Estimating Hidden Seeds
Source Institutions
In this activity, learners will estimate how many seeds are in a fruit or vegetable, then count to find out. The result: mix estimation with healthy eating.

Taste Match Game
Source Institutions
In this activity (3rd activity on the page), learners taste test different foods and categorize them as sweet, bitter, sour, or salty. Learners compare their results with the group.

¡Helados!
Source Institutions
En esta tira cómica, Mateo y Cientina emplean su conocimiento de la química y las medidas para esta actividad deliciosa - ¡hacer helado!
Mystery Writing: Write and develop a secret message
Source Institutions
Learners write an invisible message using lemon juice on a piece of paper. They then develop the message by soaking the paper in a dilute iodine solution.

Fruit Juice Mystery
Source Institutions
In this chemistry challenge, learners work to figure out which of four juices are real, and which is just food coloring and sugar.
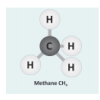
Let's Make Molecules
Source Institutions
In this activity, learners use gumdrops and toothpicks to model the composition and molecular structure of three greenhouse gases: carbon dioxide (CO2), water vapor (H2O) and methane (CH4).

Ice Cream
Source Institutions
In this chemistry activity, learners use the lowered freezing point of water to chill another mixture (ice cream) to the solid state.
