Search Results
Showing results 121 to 140 of 141

Cleaning Water with Dirt
Source Institutions
In this activity on page 7 of the PDF (Water in Our World), learners make their own water treatment systems for cleaning water.

Testing Vitamin C: Chemistry's Clear Solution
Source Institutions
In this activity on page 8 of the PDF, learners investigate vitamin C. Learners conduct a chemistry experiment to determine if Tang drink mix or orange juice contains more vitamin C.

Forwards and Backwards
Source Institutions
In this activity, learners explore acids and bases by preparing six solutions that combine vinegar and ammonia, ranging from acid (all vinegar) to base (all ammonia).

Water Treatment
Source Institutions
Water treatment on a large scale enables the supply of clean drinking water to communities.
Dirty Oil, Oily Dirt
Source Institutions
In this activity, learners discover how sediment is affected in an oil spill. Learners investigate the differences between heavy and light oil as well as the differences between different sediments.

Stick to It: Adhesion II
Source Institutions
Water sticks to all kinds of things in nature — flowers, leaves, spider webs - and doesn't stick to others, such as a duck's back.

What's in a Penny?
Source Institutions
In this chemistry activity, learners use chemical reactions to observe the composition of an alloy.

Air Pressure
Source Institutions
In this experiment, learners use a blow dryer and water bottle to observe and record changes in air pressure caused by changes in temperature.

Stuck on You: Adhesion
Source Institutions
Learners explore water adhesion and learn about why water molecules are more strongly attracted to some substances than others.
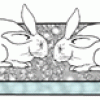
Acid Rain Eats Stone!
Source Institutions
This display shows the dangers of acid rain on buildings and other structures as two concrete bunny rabbits are disintegrated by sulfuric acid. Learners scrape chalk onto the concrete bunnies.

Squidgy Slime
Source Institutions
In this chemistry activity, learners transform two ingredients (4% polyvinyl alcohol solution and 4% borax solution) into gooey slime.

Global Climate Change and Sea Level Rise
Source Institutions
In this activity, learners practice the steps involved in a scientific investigation while learning why ice formations on land (not those on water) will cause a rise in sea level upon melting.

Having a Gas with Water
Source Institutions
In this activity, learners construct a simple electrolysis device. With this device, learners can decompose water into its elemental components: hydrogen and oxygen gas.
Light Bulb Challenge
Source Institutions
In this activity, learners explore the difference between compact fluorescent light (CFL) bulbs and traditional incandescent bulbs.
Bag of Bones
Source Institutions
In this activity, learners will use cereal to conduct an experiment and investigate how decreased bone density is related to increased risk of bone fracture.

Sunblock Investigation
Source Institutions
In this "Sid the Science Kid" activity, learners set up a simple experiment to find out how sunscreen counteracts the effects of the sun.

Forces at the Nanoscale: Nano Properties of Everyday Plants
Source Institutions
This is an activity (located on page 3 of PDF under Nasturtium Leaves Activity) about surface tension.

Soccer Kicks
Source Institutions
This activity (on page 2 of the PDF under SciGirls Activity: Soccer Ball Kick) is a full inquiry investigation into transfer of motion.

That's the Way the Ball Bounces: Level 3
Source Institutions
In this activity, learners prepare four polymer elastomers and then compare their physical properties, such as texture, color, volume, density, and bounce height.

Hot Spoons
Source Institutions
In this physics activity (page 2 of the PDF), learners will explore the concept of thermal conductivity and learn the differences between high and low thermal conductivity.
