Search Results
Showing results 61 to 78 of 78

The Colors of Flowers
Source Institutions
In this activity, learners perform an experiment to find out what determines a flower's color.

Hot Stuff!
Source Institutions
In this activity, learners discover that sand is the major ingredient in glass.

Automotive Emissions and the Greenhouse Effect
Source Institutions
In this activity about global climate change, learners will conduct an experiment and collect data to compare the amount of carbon dioxide (CO2) in four different sources of gases.

Dispersing Dispersion
Source Institutions
In this activity, learners investigate the movement caused by dispersion. Learners discover that dispersion is the random movement of objects.

Developing Tests to Distinguish Between Similar-Looking Unknowns
Source Institutions
Learners identify an unknown liquid by comparing its behavior to known liquids. Learners drop liquids onto different surfaces and see how the liquids behave.

Starch Slime
Source Institutions
Learners mix liquid water with solid cornstarch. They investigate the slime produced, which has properties of both a solid and a liquid.

Conservation of Mass
Source Institutions
This activity was designed for blind learners, but all types of learners can participate to learn about conservation of gas. This is one of the classic experiments using baking soda and vinegar.

Changing the Density of a Liquid: Heating and Cooling
Source Institutions
Learners investigate how the temperature of water affects its density.
Water Motor
Source Institutions
In this physics activity (page 10 of the PDF), learners will explore how energy from moving water can be used.
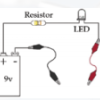
Exploring Materials: Graphene
Source Institutions
In this activity on page 4 of the PDF, explore the unique molecular structure and conductive nature of graphene. Learners construct a circuit with a battery and LED bulb.
Dirty Oil, Oily Dirt
Source Institutions
In this activity, learners discover how sediment is affected in an oil spill. Learners investigate the differences between heavy and light oil as well as the differences between different sediments.

I Can't Take the Pressure!
Learners develop an understanding of air pressure in two different activities.

"Can" You Stand the Pressure
Source Institutions
In this activity about states of matter, learners get to witness firsthand the awesome power of air pressure. They watch as an ordinary soda can is crushed by invisible forces.
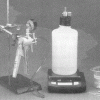
As Light as Air
Source Institutions
Learners measure a bottle full of air, and then use a vacuum pump to remove the air. When they re-weigh the bottle, learners find the mass is about 0.8g less.

Forces at the Nanoscale: Nano Properties of Everyday Plants
Source Institutions
This is an activity (located on page 3 of PDF under Nasturtium Leaves Activity) about surface tension.

Change in Temperature: Exothermic Reaction
Source Institutions
Learners add calcium chloride to a baking soda solution and observe an increase in temperature along with the production of a gas and a white precipitate. These are all signs of a chemical reaction.

Production of a Gas: Controlling a Chemical Reaction
Source Institutions
Learners mix vinegar and baking soda to produce a gas. With the addition of a bit of liquid soap, the gas becomes trapped in measurable bubbles.

Flubber: Make a polymer!
Source Institutions
This activity (on page 2 of the PDF) features a recipe to create the stretchy polymer Flubber from Borax detergent, white glue, and water.
