Search Results
Showing results 21 to 40 of 44
Ionic Bonding Puzzle Lab
Source Institutions
In this activity, learners create models of ionic compounds and observe the chemical formula of binary molecules they have created.
Rutherford's Enlarged: A Content-Embedded Activity to Teach about Nature of Science
Source Institutions
This paper describes a working-model demonstration of Ernest Rutherford's 1911 experiment about the nature of atoms.

Exploring Properties: Surface Area
Source Institutions
This hands-on activity demonstrates how a material can act differently when it's nanometer-sized.
Stretching Wires
Source Institutions
In this activity, learners determine the elastic and plastic properties of different types of metal wires.

Corals on Acid
Source Institutions
The objective of this inquiry-based lesson is for learners to gain an understanding of how increasing ocean acidity can affect the calcification of marine organisms.

Erupting Fizz
Source Institutions
This is a highly visual demonstration that illustrates both the effects of density and chemical reactions.

Lifting Lemon
Source Institutions
In this physics demonstration, learners will be surprised when a lemon slice appears to magically levitate within a pint glass.

Magical Match
Source Institutions
In this demonstration, learners will be "wowed" as three matches burn to form a triangular pyramid shape and "magically" rise off the table.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.
Gel Electrophoresis of Dyes
Source Institutions
In this experiment related to plant biotechnology, learners discover how to prepare and load an electrophoresis gel.

Taking Its Temperature
Source Institutions
In this activity (pages 5-7), learners investigate the properties of smart materials, which are materials that respond to things that happen around them.

What's the Matter
Source Institutions
In this activity, learners identify different classes of matter based on physical properties.

Extra Bounce
Source Institutions
In this indoor or outdoor demonstration, use a large and small ball to illustrate conservation of energy and momentum.
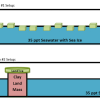
Causes and Effects of Melting Ice
Source Institutions
In this activity, learners explore the concept of density-driven currents (thermohaline circulation) and how these currents are affected by climate change.

Sand, Plants and Pants
Source Institutions
In this activity, learners explore how the application of nano-sized particles or coatings can change a bigger material’s properties.

Exploring Size: Scented Solutions
Source Institutions
This is an activity in which learners will find that they can detect differences in concentration better with their nose (smelling) than with their eyes (seeing).

Exploring Forces: Gravity
Source Institutions
In this nanoscience activity, learners discover that it's easy to pour water out of a regular-sized cup, but not out of a miniature cup.

Balloon Kebabs
Source Institutions
In this demonstration, learners observe the effects of density and pressure by attempting to make "balloon kebabs." Learners will try to insert a wooden skewer all the way through an inflated balloon
