Search Results
Showing results 41 to 60 of 216

Tinkering with Tops
Source Institutions
In this activity, learners explore the history, design and motion of spinning tops. Learners work in teams of "engineers" to design and build their own tops out of everyday items.

LEGO® Chemical Reactions
Source Institutions
This activity uses LEGO® bricks to represent atoms bonding into molecules and crystals. The lesson plan is for a 2.5 hour workshop (or four 45-minute classes).

What's In Your Breath?
Source Institutions
In this activity, learners test to see if carbon dioxide is present in the air we breathe in and out by using a detector made from red cabbage.

Making a Battery from a Potato
Source Institutions
In this electrochemistry activity, young learners and adult helpers create a battery from a potato to run a clock.

Capturing Homemade Microgravity
Source Institutions
This activity (page 2 of the PDF under SciGirls Activity: Microgravity) is a full inquiry investigation into how ordinary things behave in microgravity, similar to what astronauts experience.

My Angle on Cooling: Effects of Distance and Inclination
Source Institutions
In this activity, learners discover that one way to cool an object in the presence of a heat source is to increase the distance from it or change the angle at which it is faced.
What's So Special about Water: Absorption
Source Institutions
In this activity about water's cohesive and adhesive properties and why water molecules are attracted to each other, learners test if objects repel or absorb water.

Testing Falling Peanut Butter Sandwich Myth
Source Institutions
In this activity related to rotational inertia (page 1 of the PDF under SciGirls Activity: Microgravity), learners will use a bit of scientific experimenting to test if open-faced peanut butter sandwi
Double Dutch Distractions
Source Institutions
This activity (page 2 of the PDF under SciGirls Activity: Double Dutch) is a full inquiry investigation into whether hearing or seeing has a bigger effect on jump rope performance.

Testing for Life's Molecules
Source Institutions
In this activity, learners conduct tests for proteins, glucose, and starch.

Altered Reality
Source Institutions
In this activity, learners discover that the human brain is highly adaptable. Learners try to toss beanbags at a target while wearing prism goggles.

Crystal Gardens
Source Institutions
In this activity, which requires adult supervision, learners get to explore the awesome power of chemistry.
What Molecules Make the Holes in Bread?
Source Institutions
In this activity, learners will discover why there are holes in bread.

Water Quality and pH Levels in Aquatic Ecosystems
Source Institutions
In this fun and in depth hands-on experiment, learners test various liquid samples (distilled water, lemon juice, vinegar, and baking soda mixed with water) to determine their pH levels and identify e
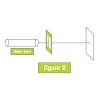
Forensic Science: Hair Sample Investigation
Source Institutions
This activity (on page 2 of the PDF under SciGirls Activity: Forensics) is a full inquiry investigation into how hairs from a crime scene are matched to suspects.

Corals and Chemistry
Source Institutions
In this activity, learners investigate how increased carbon dioxide (CO2) emissions from the burning of fossil fuels is changing the acidity (pH) of the ocean and affecting coral reefs and other marin

Glow Up
Source Institutions
In this activity, learners explore chemiluminescence and fluorescence. Learners examine 3 different solutions in regular light, in the dark with added bleach solution, and under a black light.

Air, It's Really There
Source Institutions
This lesson focuses on molecular motion in gases. Learners compare the mass of a basketball when it is deflated and after it has been inflated.

Mixtures and Solutions
Source Institutions
This activity was designed for blind learners, but all types of learners can use it to investigate heterogeneous and homogeneous mixtures and solutions, identify the differences, and explore the conce

Strong Bones, Weak Bones
Source Institutions
Most people will break a bone in their body at some point in their life, but how much force does it take to break one?
