Search Results
Showing results 21 to 40 of 50

Currently Working
Source Institutions
Learners test solutions of water, sugar, salt, and hydrochloric acid for electrical conductivity. They immerse leads from a lighting device (a battery pack connected to an LED) into each solution.
Take Out the Trash
Source Institutions
Learners explore how recyclers take advantage of the different properties of materials, such as magnetism and density, to separate them from a mixture.
Stability of Egg White Foams
Source Institutions
In this chemistry meets cooking activity, learners compare the stability of egg white foams with various additives.

Whodunit?
Source Institutions
In this fascinating and fun experiment, learners use chemistry to identify a mystery powder and to solve a "crime," a process similar to that used by real forensic scientists.
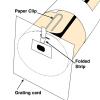
Spectroscope
Source Institutions
In this activity, learners construct their own spectroscope as they explore and observe spectra from familiar light sources.

Changing Colors
Source Institutions
In this challenge, learners have to figure out in what order to combine five solutions to change the color from clear, to yellow, to blue, and back to clear.

Fruit Juice Mystery
Source Institutions
In this chemistry challenge, learners work to figure out which of four juices are real, and which is just food coloring and sugar.
Properties of Metals
Source Institutions
In this activity, learners explore the properties of metals at four stations. The stations include A) Magnetism and Breakfast Cereal; B) Conductivity of Metals; C) Alloys; and D) Metal Plating.

Investigating Starch
Source Institutions
In this activity (on pages 10-15), learners investigate starch in human diets and how plants make starch (carbohydrates) to use as their food source.

Law of Conservation of Mass
Source Institutions
In this chemistry activity, learners explore whether matter is created or destroyed during a chemical reaction. They will compare the weight of various solutions before and after they are mixed.
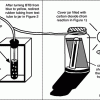
How Greenhouse Gases Absorb Heat
Source Institutions
Learners observe two model atmospheres -- one with normal atmospheric composition and another with an elevated concentration of carbon dioxide.

Blimp Jet Challenge
Source Institutions
In this design challenge activity, learners add a jet-propulsion system (i.e. a balloon) to a blimp so it flies straight and far under its own power.
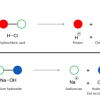
Chemical Identification
Source Institutions
In this activity, learners discover how a cabbage juice indicator helps identify acids and bases, and how iodine indicates the presence of starch.

Disappearing Colors
Source Institutions
In this challenge, learners figure out how to make a juice stain disappear.
Hot and Cold: Endothermic and Exothermic Reactions
Source Institutions
Visitors mix urea with water in one flask and mix calcium chloride with water in another flask. They observe that the urea flask gets cold and the calcium chloride flask gets hot.

Model Wind Tunnel
Source Institutions
In this activity, learners build a miniature wind tunnel to measure force. Learners construct the model out of Lexan plastic, a fan, and a precise digital scale.
It's A Gas!
Source Institutions
Visitors mix water and sodium bicarbonate (baking soda) in a large flask. They then add citric acid to the mixture and stopper the flask. The resulting reaction creates carbon dioxide gas.
All Mixed Up!: Separating Mixtures
Source Institutions
Visitors separate a mixture of pebbles, salt crystals, and wood shavings by adding water and pouring the mixture through a strainer.
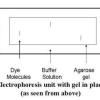
Gel Electrophoresis of Dyes
Source Institutions
In this experiment related to plant biotechnology, learners discover how to prepare and load an electrophoresis gel.

Photosynthetic Pictures Are Worth More Than a Thousand Words
Source Institutions
This activity provides an opportunity for learners to observe and examine how carbon dioxide, water, and light produce glucose/starch through a process called photosynthesis.
