Search Results
Showing results 321 to 340 of 799

Mighty Molecules
Source Institutions
In this activity, learners use marshmallows and gum drops to construct seven models of molecules. Learners classify (solid, liquid or gas) and draw diagrams of the molecules.
Smell Detective
Source Institutions
In this activity about olfaction (8th activity on the page), learners smell 10 different items with different odors. Then they try to identify the smells when they are mixed together.

Science of Sunblock
Source Institutions
This is an activity (located on page 3 of the PDF under Stained Glass Activity) about nanotechnology making its way into everyday products, such as sunscreen, and how effective these invisible particl

Comparing Crystals
Source Institutions
In this chemistry activity (page 3 of the PDF), learners will learn about crystals by growing their very own.
Chemical Reactions in Your Mouth
Source Institutions
In this chemistry activity (page 5 of the PDF), learners will see that chewing is more than just the crushing up of food; there is actually a chemical change going on at the same time.

Temperature vs. Height: Soda Geyser Series #6
Source Institutions
In this activity, learners conduct a controlled experiment to examine how temperature will affect the height of a soda geyser.
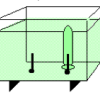
Electrolysis
Source Institutions
Using electrolysis, learners produce hydrogen gas and oxygen gas from water molecules in a solution.

Cabbage Juice Indicator: Test the pH of household products
Source Institutions
Learners make their own acid-base indicator from red cabbage. They use this indicator to test substances around the house.

Shrinkers: Cook up some plastic!
Source Institutions
In this activity (on page 2 of the PDF), learners (with adult help and supervision) investigate how heat affects polystyrene plastic.

Gassy Lava Lamp
Source Institutions
In this activity, learners use oil, water, food coloring and antacid tablets to create a bubbling lava lamp. Use this activity to introduce concepts related to density, hydrophobicity vs.
Choose Your Ooze
Source Institutions
During this activity, learners will make different versions of "ooze" using varied proportions of detergent and glue.

Miscibility
Source Institutions
Learners observe a bottle containing water and oil. They are invited to pick up the bottle and mix the contents together.

Potato Power
Source Institutions
Learners combine hydrogen peroxide with three different forms of potato: raw chunks, ground chunks, and boiled chunks.

Energy Sources
Source Institutions
In this activity about the relationship between food and energy (page 5 of PDF), learners conduct an experiment to compare how much energy is released as heat from two different foods.
Pollution Diffusion
Source Institutions
Learners design their own experiment to investigate how pollution diffuses through ground material.

Currently Working
Source Institutions
Learners test solutions of water, sugar, salt, and hydrochloric acid for electrical conductivity. They immerse leads from a lighting device (a battery pack connected to an LED) into each solution.
Milli's Super Sorting Challenge
Source Institutions
In this activity, learners separate materials based on their special properties to mimic the way recyclables are sorted at recycling centers.

Glitter Bottles
Source Institutions
In this activity, learners will create glitter bottles with adult supervision. These are a fun way for little ones to practice observing and making comparisons.

Column Chromatography
Source Institutions
In this activity, learners separate the components of Gatorade using a home-made affinity column.
Outrageous Ooze: Is It a Liquid or a Solid?
Source Institutions
This activity provides instructions for using cornstarch and water to make an ooze which has the properties of both a solid and liquid.
